295,00 € – 895,00 €
Product details
Synonyms = VIL1; Villin-1; Villin1
Antibody type = Recombinant Rabbit monoclonal / IgG
Clone = MSVA-459R
Positive control = Colon: A moderate to strong villin staining with predominance of the apical membrane should be seen in all epithelial cells. Liver: A weak to moderate staining of the apical pole of hepatocytes should be seen.
Negative control = Colon: Villin staining should be absent in all non-epithelial cells.
Cellular localization = Cell Surface and Cytoplasmic
Reactivity = Human
Application = Immunohistochemistry
Dilution = 1:100 – 1:200
Intended Use = Research Use Only
Relevance of Antibody
Biology Behind
Villin is a 92.5 kDa protein encoded by the VIL1 gene located at the human chromosome 2q35. The protein is mainly localized in the microvilli of the brush border of various normal epithelial cell types. As an actin-binding protein, Villin is connected to the actin core bundle of the brush border. Villin has actin modifying functions and is involved in the nucleation, crosslinking, capping and splitting of actin filaments. Amongst others, the actin modifying functions are regulated by calcium and phosphorylation of the villin protein. Functional studies suggest an impact of Villin upregulation on the cytoskeleton architecture, cellular shape and motility as well as cell viability.
Staining Pattern in Normal Tissues
Villin staining pattern in Normal Tissues with antibody MSVA-459R (images are shown in our “Normal Tissue Gallery”)
| Brain | Cerebrum | Negative. |
| Cerebellum | Negative. | |
| Endocrine Tissues | Thyroid | Negative. |
| Parathyroid | Negative. | |
| Adrenal gland | Negative. | |
| Pituitary gland | Negative. | |
| Respiratory system | Respiratory epithelium | Negative. |
| Lung | Negative. | |
| Gastrointestinal Tract | Salivary glands | Negative. |
| Esophagus | Negative. | |
| Stomach | Weak to moderate villin positivity of surface epithelium. Gradual decrease of staining intensity towards the base of the glands. | |
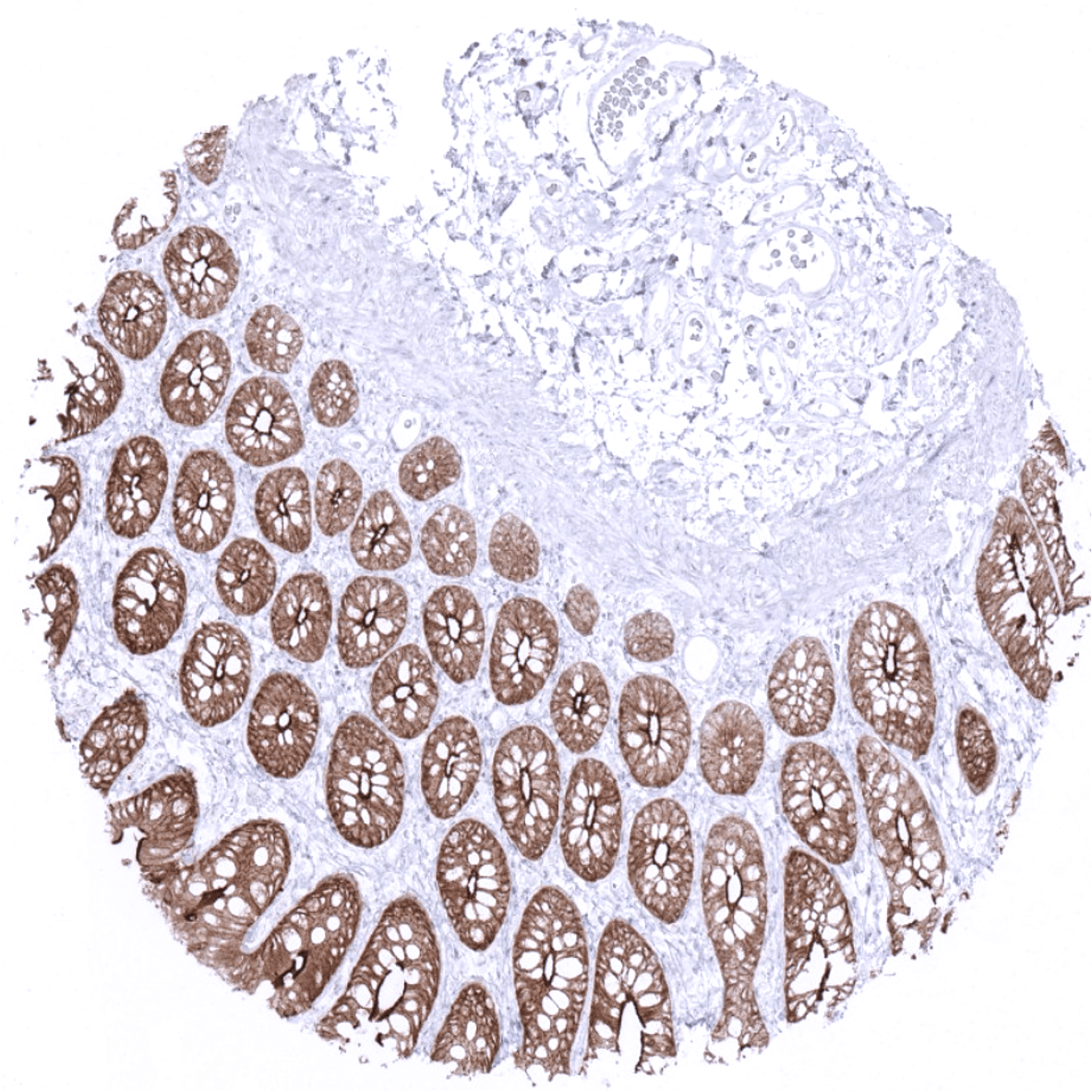
| Duodenum | Strong villin positivity in all epithelial cells. | |
| Small intestine | Strong villin positivity in all epithelial cells. | |
| Appendix | Strong villin positivity in all epithelial cells. | |
| Colon | Strong villin positivity in all epithelial cells. | |
| Rectum | Strong villin positivity in all epithelial cells. | |
| Liver | Weak to moderate villin staining of luminal membranes of hepatocytes (zonal variability of staining intensity). | |
| Gallbladder | Negative. | |
| Pancreas | Strong villin positivity in excretory ducts. Weak to moderate villin staining of luminal membranes of acinar cells. | |
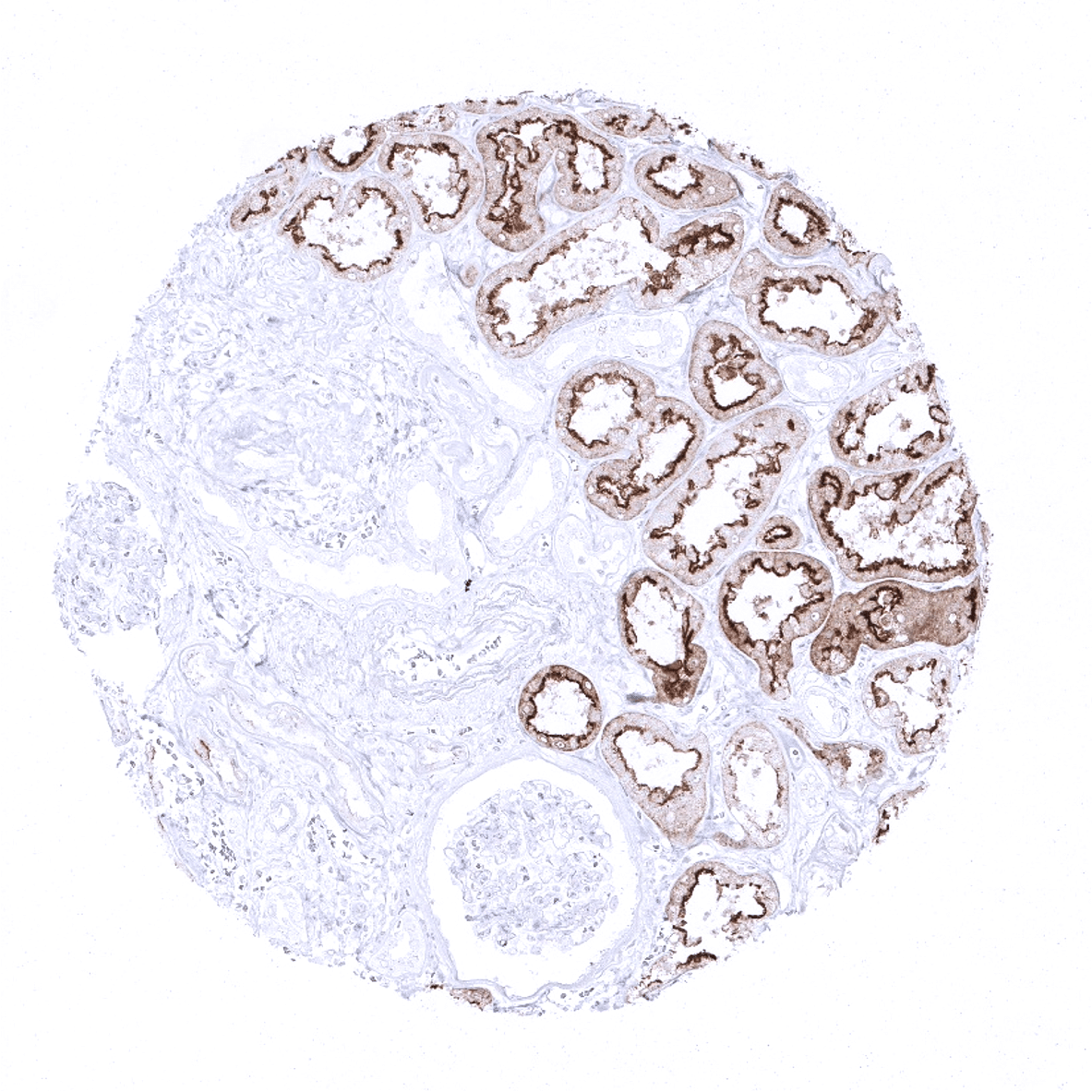
| Genitourinary | Kidney | Strong villin positivity in proximal tubuli. |
| Urothelium | Negative. | |
| Male genital | Prostate | Negative. |
| Seminal vesicles | Negative. | |
| Testis | Negative. | |
| Epididymis | Weak to moderate villin staining of ciliated columnar cells of the cauda. | |
| Female genital | Breast | Negative. |
| Uterus, myometrium | Negative. | |
| Uterus, ectocervix | Negative. | |
| Uterus endocervix | Negative. | |
| Uterus, endometrium | Negative. | |
| Fallopian Tube | Negative. | |
| Ovary | Negative. | |
| Placenta early | Negative. | |
| Placenta mature | Negative. | |
| Amnion | Negative. | |
| Chorion | Negative. | |
| Skin | Epidermis | Negative. |
| Sebaceous glands | Negative. | |
| Muscle/connective tissue | Heart muscle | Negative. |
| Skeletal muscle | Negative. | |
| Smooth muscle | Negative. | |
| Vessel walls | Negative. | |
| Fat | Negative. | |
| Stroma | Negative. | |
| Endothelium | Negative. | |
| Bone marrow/ lymphoid tissue | Bone marrow | Negative. |
| Lymph node | Negative. | |
| Spleen | Negative. | |
| Thymus | Negative. | |
| Tonsil | Negative. | |
| Remarks | Villin staining is usually membranous but is also seen in the cytoplasm. It often predominates on luminal membranes. |
Findings obtained by MSVA-459R are largely consistent to the RNA data summarized in the Human Protein Atlas (Tissue expression Villin). The MSVA-459R findings confirm that the most significant staining occurs in the distal gastrointestinal tract but show that a weak staining of surface epithelium also occurs in the stomach. In addition, our documentation describes weak to moderate villin expression in the epididymis caput that was not previously identified by RNA screening and assigns RNA expression data to specific cell types with protein expression such as acinar and ductal epithelial cells in the pancreas.
Suggested positive tissue control: Colon: A moderate to strong staining with predominance of the apical membrane should be seen in all epithelial cells. Liver: A weak to moderate staining of the apical pole of hepatocytes should be seen.
Suggested negative tissue control: Colon: Villin staining should be absent in all non-epithelial cells.
Staining Pattern in Relevant Tumor Types
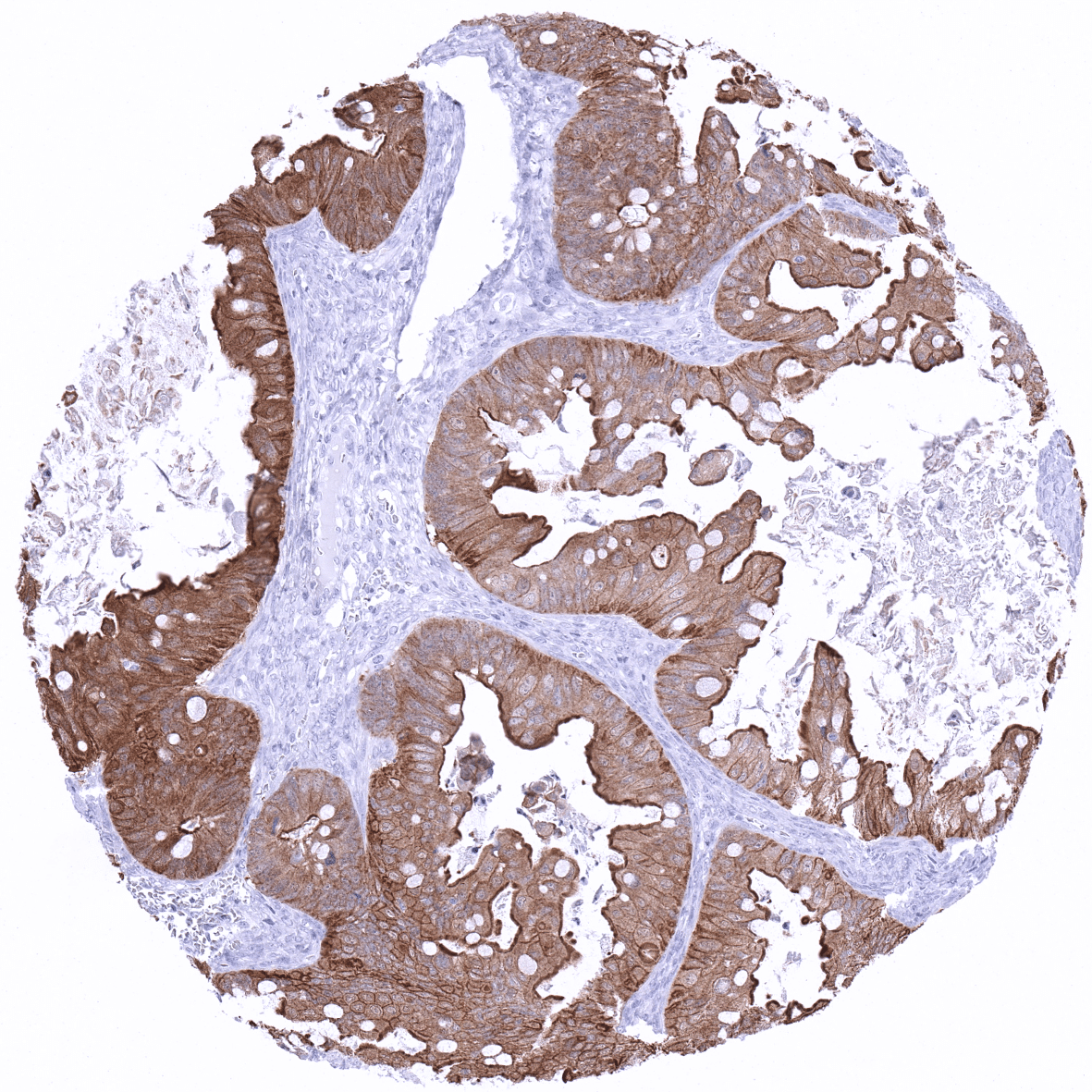
In carcinomas, villin expression is predominantly cytoplasmic. Villin positivity is most commonly seen in colorectal cancer. Tumors that were described to also show villin expression in a variable fraction of cases include gastric, duodenal and esophageal carcinoma, gastrointestinal neuroendocrine tumors, endometrial carcinoma, hepatocellular carcinoma, pulmonary adenocarcinoma (enteric type) and others.
The TCGA findings on Villin RNA expression in different tumor categories have been summarized in the Human Protein Atlas.
Compatibility of Antibodies
Villin (MSVA-459R) publication summary
Relevant publication: Dum et al.“Villin expression in human tumours: a tissue microarray study on 14,398 tumours”. Published in Expert Review of Molecular Diagnostics Accepted 18 Jul 2022, Accepted author version posted online: 22 Jul 2022, PMID: 35866621
A total of 12,429 tumors were successfully analyzed from 118 different tumor categories by using the following protocol: Heat-induced antigen retrieval for 5 minutes in an autoclave at 121°C in pH7,8 Target Retrieval Solution buffer. MSVA-459R at a dilution of 1:250 at 37°C for 60 minutes. Visualization of bound antibody by the EnVision Kit (Dako, Agilent). This protocol was also used for all stainings depicted in our tumor and normal tissue galleries.
In this study, at least one positive case was seen in 54 (46%) of 118 tumor categories and 36 (31%) tumor categories included at least one case with strong positivity The highest frequencies of villin positivity (and the highest levels of expression) were seen in adenocarcinomas and neuroendocrine tumors of the colorectum (60-100%), the upper gastrointestinal tract (61-100%), and of the pancreatobiliary system (25-86%). Villin positivity was also frequent in yolk sac tumors of the testis (76%) and in mucinous carcinomas of the ovary (67%), various categories of extra-gastrointestinal neuroendocrine neoplasms (22-41%), kidney tumors (up to 18%), hepatocellular carcinomas (25%), endometroid carcinomas of the ovary (20%) and of the endometrium (12%). Villin staining – mostly weak – was seen in only 12% of adenocarcinomas of the lung. The distribution of positive staining results is shown in an “organ-systematic” and in a “ranking order” figure below (images based on data from Dum et al.). Results on possible associations with histopathological and clinical parameters of tumor aggressiveness are also summarized below (table based on data from Dum et al.).
Authors conclusions on diagnostic utility of villin immunohistochemistry with respect to the distinction of benign versus malignant (Dum et al.):
- not applicable.
Authors conclusions on diagnostic utility of villin immunohistochemistry with respect to the distinction of different tumor entities (Dum et al.):
- Distinction of yolk sac tumors of the testis (mostly positive) from other neoplasms of the testis (usually negative).
- Distinction of metastases from gastrointestinal neoplasms (usually positive) from pulmonary adenocarcinoma (mostly negative).
- Villin immunohistochemistry should be applied as a part of a marker panel rather than as a stand-alone marker.
- Importantumor entities that are nearly always negative: prostate cancer, breast cancer, serous ovarian cancer, urothelial carcinoma, mesothelioma, thyroid cancer.
Authors conclusions on prognostic/predictive role of villin expression (Dum et al.):
- In colorectal adenocarcinomas, reduced villin expression was significantly linked to unfavorable tumor phenotype and microsatellite instability.
- In clear cell renal cell carcinomas, low villin expression was linked to high grade (p<0.04), advanced pT stage (p=0.0044), and distant metastasis (p=0.0072).
Data from the publication: “Villin expression in human tumours: a tissue microarray study on 14,398 tumours”. Published by Dum et a. in Expert Review of Molecular Diagnostics Accepted 18 Jul 2022, Accepted author version posted online: 22 Jul 2022, PMID: 35866621
Figure 1. Villin staining in cancer (“organ-systematic”; according to Dum et al.)
Figure 2. Villin staining in cancer (“ranking list”; according to Dum et al.)
Figure 3. Clinico-pathological associations described by Dum et al. (p-value)
Protocol Recommendations
IHC users have different preferences on how the stains should look like. Some prefer high staining intensity of the target stain and even accept some background. Others favor absolute specificity and lighter target stains. Factors that invariably lead to more intense staining include higher concentration of the antibody and visualization tools, longer incubation time, higher temperature during incubation, higher temperature and longer duration of the heat induced epitope retrieval (slide pretreatment). The impact of the pH during slide pretreatment has variable effects and depends on the antibody and the target protein. Accordingly, multiple different protocols can generate identical staining results.
All images and data shown here and in our image galleries are obtained by the manual protocol described below. Other protocols resulting in equivalent staining are described as well.
-Manual protocol
Freshly cut sections should be used (less than 10 days between cutting and staining). Heat-induced antigen retrieval for 5 minutes in an autoclave at 121°C in pH 9 Target Retrieval Solution buffer. Apply MSVA-459R at a dilution of 1:125 at 37°C for 60 minutes. Visualization of bound antibody by the EnVision Kit (Dako, Agilent) according to the manufacturer’s directions
-Impact of pH
MSVA-459R results in strongest staining if pH9,0 is used for slide pretreatment. pH7,8 is acceptable but lower pH results in a significant reduction of sensitivity.
Potential Research Applications
The clinical/prognostic significance of villin expression in individual tumor types is unknown.
Evidence for Antibody Specificity in IHC
Specificity of MSVA-459R is documented by strong positive staining in cell types that are well documented to express villin such as epithelial cells of the colorectum, appendix, small intestine, duodenum, pancreatic excretion ducts and in proximal tubuli of the kidney as well as absence of staining in all tissues known to not express villin including tissues notorious for non-specific IHC background such squamous epithelium or distal tubuli of the kidney.
The validity of the normal tissue staining patterns is also confirmed by a comparison of MSVA-459R staining with staining results obtained by a second independent, commercially available antibody (RTU IVD).
Antibody comparison: MSVA-459R vs another commercial anti-Villin antibody RTU (IVD)