295,00 € – 995,00 €
Product details
Synonyms = Migration inducing gene 9 (MIG9); S100E; S100 calcium binding protein P
Antibody type = Recombinant Rabbit monoclonal / IgG
Clone = MSVA-480R
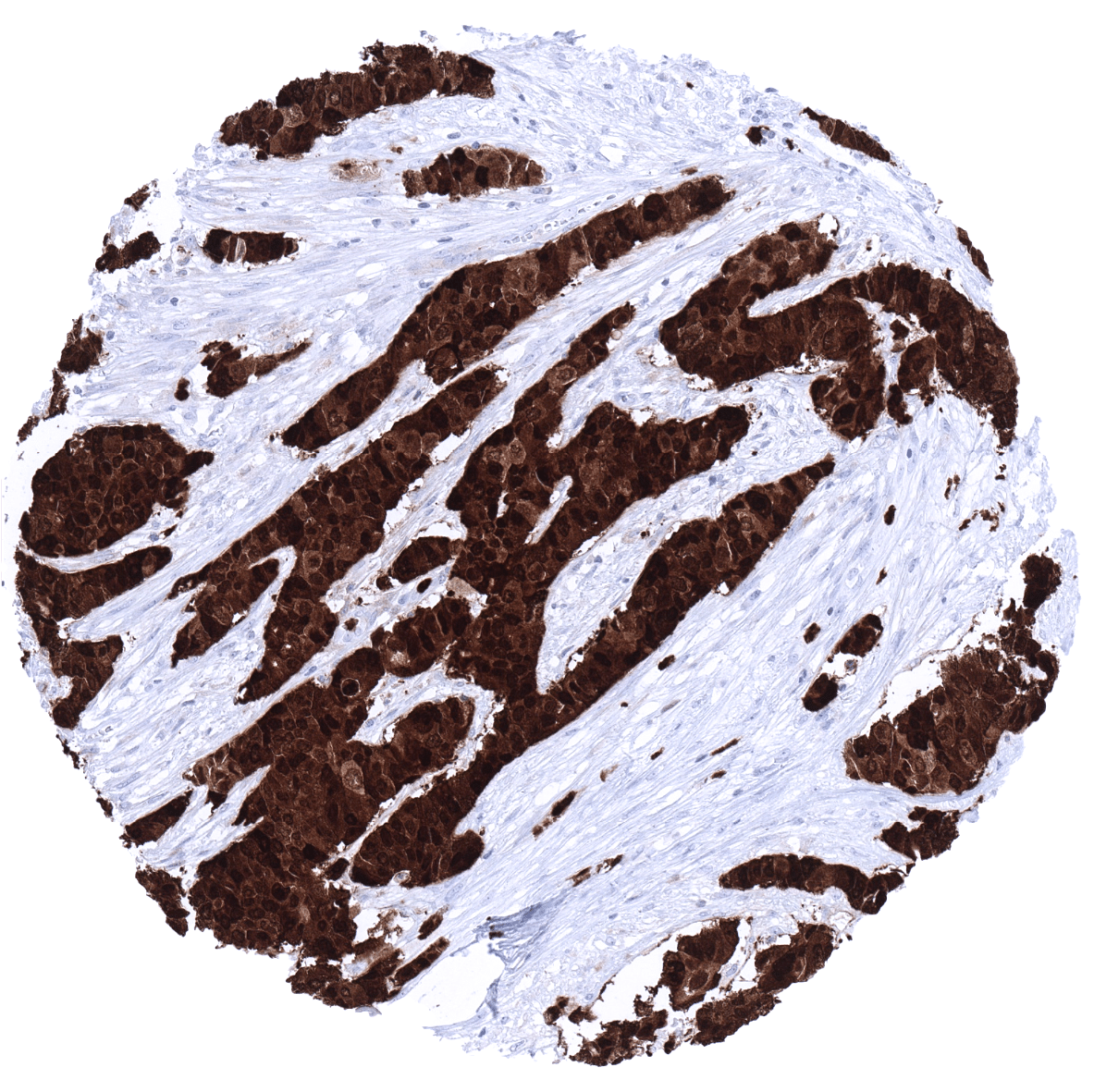
Positive control = Urinary bladder: A strong S100P immunostaining should be seen in the urothelium but not in the stroma or muscular cells.
Negative control = Kidney: S100P immunostaining should be completely absent.
Cellular localization = Nucleus and cytoplasm
Reactivity = Human
Application = Immunohistochemistry
Dilution = 1:100-200
Intended Use = Research Use Only
Relevance of Antibody
S100P is expressed in placenta, urothelium, stomach, and respiratory epithelium.
Biology Behind
S100P is a 10.4 kDa protein, coded by the S100P gene at 4p16. The S100 genes are a group of water soluble low-molecular-weight (9–12 kD) proteins characterized by two calcium-binding sites that have a specific helix-loop-helix (“EF-hand type”) conformation. The “S100” gene name is derived from the fact that these proteins are soluble in 100%. S100P interacts, directly or indirectly, with a number of different proteins, many of which regulate actin cytoskeleton dynamics and extracellular matrix remodeling. Interaction partners for example include Ezrin, IQGAP1, myosin IIA, cathepsin D, and cofilin. Through these interactions, S100P integrates and regulates various signaling pathways and induces a broad range of important functional results.
Staining Pattern in Normal Tissues
A strong S100P immunostaining is regularly seen in trophoblastic (syncytiotrophoblast more than cytotrophoblast) and chorion cells of the placenta, all cell layers of urothelium (stronger intensity of the top layers than in the basal layers), surface epithelial cells and neck cells (but not of glands) of the stomach, and in granulocytes. A weak to moderate S100P staining can be found in variable fractions of epithelial cells of the colon mucosa (stronger in superficial than in crypt cells), duodenum and jejunum (mainly in goblet cells), transitional epithelium of the anal canal, as well as in mucinous and basal cells of sublingual (but not submandibular) glands. In the tonsil crypts, a weak to moderate S100P staining occurs in the superficial layers of squamous epithelium. In the thymus, a moderate staining of corpuscles of Hassall’s but not of other epithelial cells is seen. S100P can occasionally also occur in several other normal tissues, perhaps due to specific functional changes. These tissues include gallbladder epithelium (in one sample with considerable inflammation), respiratory epithelium of bronchi and paranasal sinus (non-basal cell types), and endometrium (especially in secretion phase). S100P immunostaining is absent in lung, liver, pancreas, prostate, testis, kidney, Brunner glands, fallopian tube, endocervix, adrenal gland, thyroid, parathyroid gland, adeno- and neurohypophysis, lymph node, and mesenchymal tissues. A fibrillar staining observed in the white matter of the cerebellum may represent a (tolerable) cross-reactivity.
These findings are largely consistent with the RNA and protein data described in the Human Protein Atlas (Tissue expression S100P)
Positive control: Urinary bladder: A strong S100P immunostaining should be seen in the urothelium but not in the stroma or muscular cells.
Negative control: Kidney: S100P immunostaining should be completely absent.
Staining Pattern in Relevant Tumor Types
S100P immunostaining is particularly strong and frequent in urothelial, colorectal and gastrointestinal tumors. Various other tumor entities have been described to express S100P in a fraction of cases.
The TCGA findings on S100P RNA expression in different tumor categories have been summarized in the Human Protein Atlas.
Compatibility of Antibodies
No data available at the moment
Protocol Recommendations
IHC users have different preferences on how the stains should look like. Some prefer high staining intensity of the target stain and even accept some background. Others favor absolute specificity and lighter target stains. Factors that invariably lead to more intense staining include higher concentration of the antibody and visualization tools, longer incubation time, higher temperature during incubation, higher temperature and longer duration of the heat induced epitope retrieval (slide pretreatment). The impact of the pH during slide pretreatment has variable effects and depends on the antibody and the target protein.
All images and data shown here and in our image galleries are obtained by the manual protocol described below. Other protocols resulting in equivalent staining are described as well.
Manual protocol
Freshly cut sections should be used (less than 10 days between cutting and staining). Heat-induced antigen retrieval for 5 minutes in an autoclave at 121°C in pH 7,8 Target Retrieval Solution buffer. Apply MSVA-480R at a dilution of 1:150 at 37°C for 60 minutes. Visualization of bound antibody by the EnVision Kit (Dako, Agilent) according to the manufacturer’s directions.
Agilent / Dako – Autostainer Link 48
Pretreatment in PT-Link for 30 minutes at 95°C (pH high); FLEX peroxidase blocking for 5 minutes (room temperature), MSVA-480R 1:150 for 20 minutes (room temperature), FLEX+ mouse/rabbit (LINKER) for 15 minutes (room temperature), horseradish peroxidase (HRP) for 20 minutes (room temperature), FLEX DAB+Sub-Chromo for 10 minutes (room temperature), FLEX hematoxylin for 5 minutes (room temperature).
These images reflect stainings by the protocol described above. It is of note that a comparable staining result can also be obtained by different protocols. In general, a longer pretreatment, a longer incubation time of the primary antibody, a higher antibody concentration, and a longer incubation time of FLEX+LINKER result in stronger staining, potentially at the cost of more background staining. Modifications of the protocol with a strengthening effect on staining intensity in combination with changes of other parameters that result in lower staining intensity can result in a comparable result as shown above.
Leica – BOND RX
Dewax at 72°C for 30 seconds; Pretreatment in Bond Epitope Retrieval Solution (ER2 – EDTA pH9) for 20 minutes at 100°C; Peroxidase blocking for 5 minutes (room temperature), MSVA-480R 1:150 for 15 minutes (room temperature), Post primary (rabbit anti mouse) for 8 minutes (room temperature), Polymer (goat anti rabbit) for 8 minutes (room temperature), mixed DAB refine for 10 minutes (room temperature), hematoxylin for 5 minutes (room temperature).
These images reflect stainings by the protocol described above. It is of note that a comparable staining result can also be obtained by different protocols. In general, a longer pretreatment, a longer incubation time of the primary antibody, a higher antibody concentration, a higher temperature during incubation, and a longer incubation time of Post primary and or the Polymer result in stronger staining, potentially at the cost of more background staining. Modifications of the protocol with a strengthening effect on staining intensity in combination with changes of other parameters that result in lower staining intensity can result in a comparable result as shown above.
Roche – Ventana Discovery ULTRA
Pretreatment for 64 minutes at 100°C (pH 8,4); CM peroxidase blocking for 12 minutes (room temperature), MSVA-480R 1:150 for 20 minutes at 36°C, secondary antibody (anti-rabbit HQ) for 12 minutes at 36°C, anti-HQ HRP for 12 minutes at room temperature, DAB at room temperature, hematoxylin II at room temperature for 8 minutes, bluing reagent at room temperature for 4 minutes.
These images depict staining results obtained by the protocol described above. It is of note, that the Ventana machines generally require higher antibody concentrations than other commonly used autostainers because the antibodies are automatically diluted during the procedure. Various other protocols can result in an identical result as shown above. A longer pretreatment, a longer incubation time of the primary antibody, a higher antibody concentration, a higher temperature during incubation, and a longer incubation time of secondary antibody and or the anti-HQ HRP result in stronger staining, potentially at the cost of more background staining.
Potential Research Applications
- The diagnostic utility of S100 expression analysis should be further investigated in a large cohort of tumors from different entities
- The diagnostic role of S100P as a parameter of malignancy needs to be investigated.
- The prognostic role of S100P expression in tumors is not sufficiently analyzed so far.
- The role of S100P in inflammatory disease should be further evaluated.
Evidence for Antibody Specificity in IHC
There are two ways how the specificity of antibodies can be documented for immunohistochemistry on formalin fixed tissues. These are: 1. comparison with a second independent method for target expression measurement across a large number of different tissue types (orthogonal strategy), and 2. Comparison with one or several independent antibodies for the same target and showing that all positive staining results are also seen with other antibodies for the same target (independent antibody strategy).
Orthogonal validation: For the antibody MSVA-480R specificity is suggested by the strong concordance of the immunostaining data with data from three independent RNA screening studies, including the Human Protein Atlas (HPA) RNA-seq tissue dataset, the FANTOM5 project, and the Genotype-Tissue Expression (GTEx) project, which are all summarized in the Human Protein Atlas (Tissue expression S100P). Immunostaining by using MSVA-480R was almost exclusively detected in organs and cell types with documented RNA expression such as placenta, stomach, urinary bladder, colon, tonsil, and spleen (granulocytes). Low to moderate level RNA expression have also been described for the lung, prostate, esophagus and the skin which were not confirmed in an immunohistochemical normal tissue analysis by using MSVA-480R. It appears possible that RNA derived from respiratory epithelium (lung), granulocytes (skin), urothelium (prostate) or stomach (esophagus) may have caused the detection of S100P RNAs in these tissues.
Tissues in which S100P immunostaining could not be confirmed by RNA data included endometrium, gallbladder, duodenum/jejunum (only very few cells S100P IHC positive) for which the RNA analyses did not show S100P expression as well as transitional epithelium of the anal canal, thymus, respiratory epithelium and sublingual glands. For these latter tissues, accessible RNA data do not exist.
Comparison of antibodies: True expression of S100P in respiratory epithelium and endometrium was confirmed by the independent antibody HPA019502 used for protein analyses in the protein atlas.
Some other normal tissue stainings obtained by MSVA-480R were not confirmed by HPA019502 either because the respective tissues were not analyzed in the protein atlas (thymus, transitional epithelium of the anal canal, thymus, sublingual glands) or because the few tissues analyzed in the protein atlas were negative (gallbladder, duodenum/jejunum). The latter finding is not unexpected because these tissues do only occasionally show S100P staining by MSVA-480R, most likely in case of specific functional alterations as for example in a case of a heavily inflamed gallbladder sample.
A fibrillar staining observed in the white matter of the cerebellum may represent a (tolerable) cross-reactivity because it was not reproduced by HPA019502.