295,00 € – 995,00 €
Product details
Synonyms = NCL; Nucl; Nucleolin; Protein C23
Antibody type = Recombinant Rabbit monoclonal / IgG
Clone = MSVA-623R
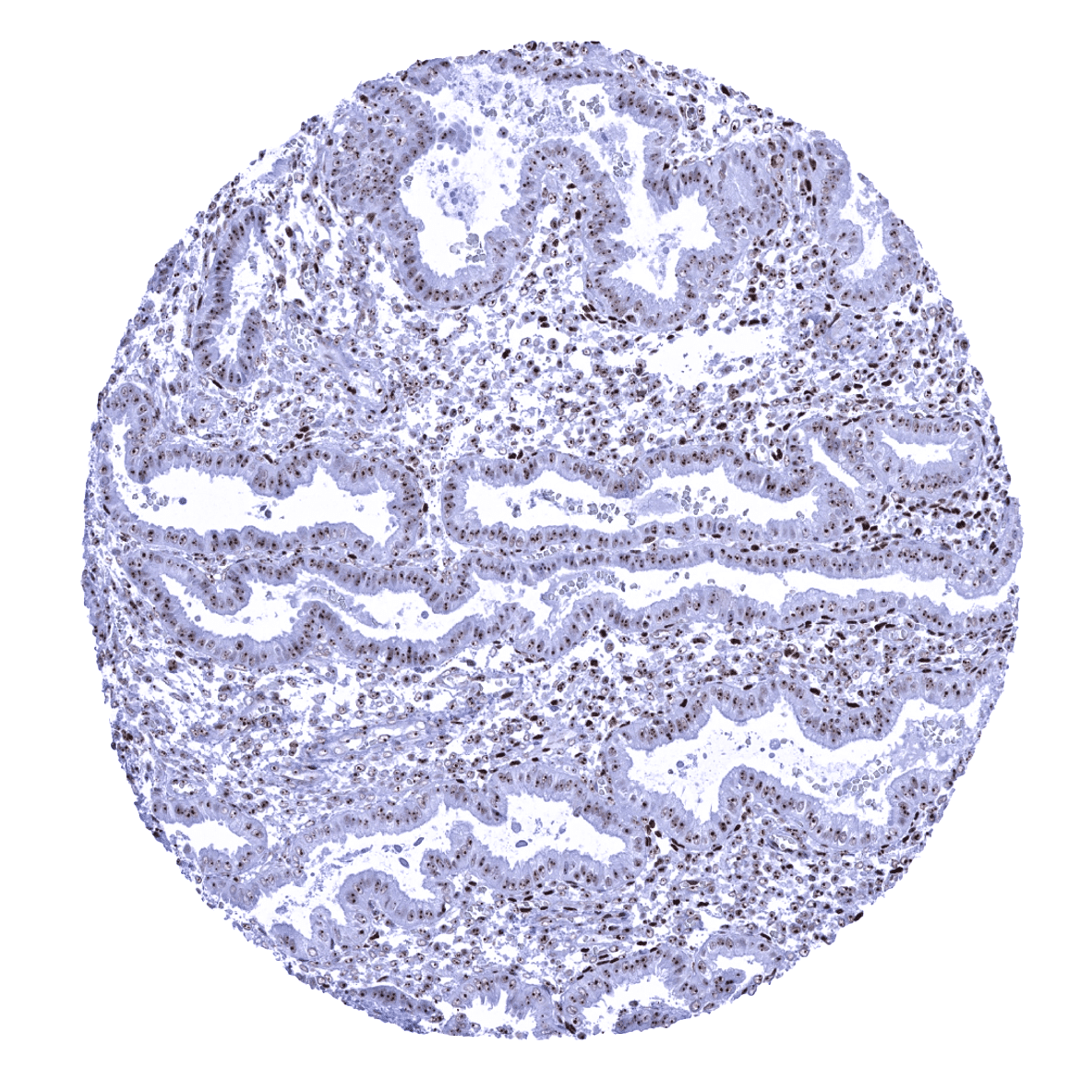
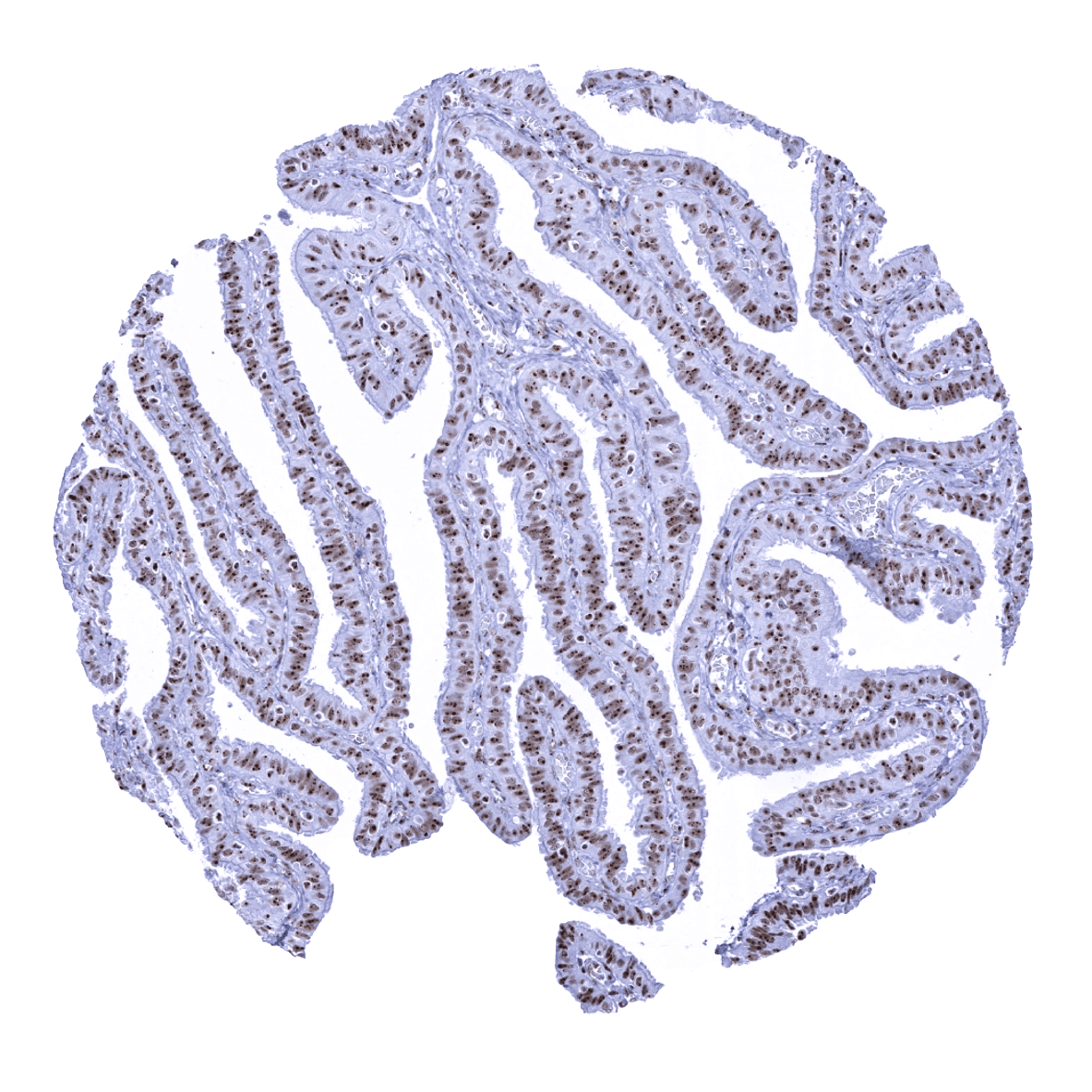
Positive control = Colon: A nuclear nucleolin immunostaining should be seen in all cells. A focus of the staining on nucleoli should be visible – at least – in the superficial epithelial cell layer.
Negative control = Colon: A cytoplasmic or membranous nucleolin immunostaining should be absent in all cells.
Cellular localization = Nuclear.
Reactivity = Human
Application = Immunohistochemistry
Dilution = 1:100-200
Intended Use = Research Use Only
Relevance of Antibody
Nucleolin is a pivotal protein for transcriptional regulation located in nuclei and nucleoli
Biology Behind
Nucleolin (NCL) also termed C23 is a 100-kDa multifunctional DNA/RNA-binding nucleolar phosphoprotein coded by the NCL gene located at chromosome 2q37.1. Nucleolin is one of the most common proteins in the nucleolus and accounts for about 10% of the protein content in the nucleolus. It is highly expressed in proliferating cells and involved in ribosome assembly, transcriptional regulation, chromatin remodeling, cell proliferation, differentiation, and apoptosis. Nucleolin binds a broad variety of nucleic acids and induces chromatin decondensation by binding to histone H1. Nucleolin can also be transferred to the cell membrane, where it, for example, acts as a receptor for the Respiratory Syncytial Virus (RSV) fusion protein. Nucleolin plays an important role in cancer where increased levels of nucleolin can be found in the nuclei, and the level of cytoplasmic and cell surface nucleolin may be increased. Nucleolin depletion leads to cell cycle arrest. Both nucleolar and cell surface nucleolin are considered potential therapeutic targets.
Staining Pattern in Normal Tissues
Nucleolin occurs in virtually all nuclei of all cells in all tissues. In cell types with discernible nucleoli, the immunostaining is most distinct in nucleoli. In other tissues, the entire nucleus is homogeneously stained. Slight variations of the staining intensity occur between tissues and cell types. In general, staining intensity appears highest in tissues with high proliferation rate. Nucleolin staining can be lower in some tissues with particularly low proliferation such as gastric glands, superficial epithelium of the colon, decidua cells, or neurons. Cytoplasmic and/or membranous nucleolin immunostaining is not seen in normal striated muscle, heart muscle, smooth muscle, myometrium of the uterus, corpus spongiosum of the penis, ovarian stroma, fat, skin (including hair follicle and sebaceous glands), oral mucosa of the lip, oral cavity, surface epithelium of the tonsil, and transitional mucosa of the anal canal, ectocervix, squamous epithelium of the esophagus, urothelium of the renal pelvis and urinary bladder, decidua, placental trophoblastic cells, lymph node, spleen, thymus, tonsil, mucosa of the stomach, duodenum, ileum, appendix, colon, rectum and gall bladder, pancreas, liver, parotid gland, submandibular gland, sublingual gland, Brunner gland of the duodenum, cortex and medulla of the kidney, prostate, seminal vesicle, epididymis, testis, respiratory epithelium and glands of bronchi and paranasal sinus, lung, breast, endocervix, endometrium, fallopian tube, corpus luteum and follicular cyst of the ovary, adrenal gland, parathyroid gland, cerebellum, cerebrum and pituitary gland.
These findings are largely consistent with the RNA and protein data described in the Human Protein Atlas (Tissue expression Nucleolin)
Positive control: Colon: A nuclear nucleolin immunostaining should be seen in all cells. A focus of the staining on nucleoli should be visible – at least – in the superficial epithelial cell layer.
Negative control: Colon: A cytoplasmic or membranous nucleolin immunostaining should be absent in all cells.
Staining Pattern in Relevant Tumor Types
A nuclear and nucleolar immunostaining is seen in virtually all tumors while the intensity of the staining is variable to some extent. In a fraction of tumors, the staining is not purely nuclear but also cytoplasmic.
The TCGA findings on Nucleolin RNA expression in different tumor categories have been summarized in the Human Protein Atlas.
Compatibility of Antibodies
No data available at the moment
Protocol Recommendations
IHC users have different preferences on how the stains should look like. Some prefer high staining intensity of the target stain and even accept some background. Others favor absolute specificity and lighter target stains. Factors that invariably lead to more intense staining include higher concentration of the antibody and visualization tools, longer incubation time, higher temperature during incubation, higher temperature and longer duration of the heat induced epitope retrieval (slide pretreatment). The impact of the pH during slide pretreatment has variable effects and depends on the antibody and the target protein.
All images and data shown here and in our image galleries are obtained by the manual protocol described below. Other protocols resulting in equivalent staining are described as well.
Manual protocol
Freshly cut sections should be used (less than 10 days between cutting and staining). Heat-induced antigen retrieval for 5 minutes in an autoclave at 121°C in pH 7,8 Target Retrieval Solution buffer. Apply MSVA-623R at a dilution of 1:150 at 37°C for 60 minutes. Visualization of bound antibody by the EnVision Kit (Dako, Agilent) according to the manufacturer’s directions.
Agilent / Dako – Omnis
Pretreatment in PT-Link for 30 minutes at 95°C (pH high); FLEX peroxidase blocking for 5 minutes (room temperature), MSVA-623R 1:150 for 20 minutes at 36°C, FLEX+ mouse/rabbit (LINKER) for 10 minutes at 36°C, horseradish peroxidase (HRP) for 20 minutes (room temperature), FLEX DAB+Sub-Chromo for 10 minutes (room temperature), FLEX hematoxylin for 5 minutes (room temperature).
These images reflect stainings by the protocol described above. It is of note that a comparable staining result can also be obtained by different protocols. In general, a longer pretreatment, a longer incubation time of the primary antibody, a higher antibody concentration, and a longer incubation time of FLEX+LINKER result in stronger staining, potentially at the cost of more background staining. Modifications of the protocol with a strengthening effect on staining intensity in combination with changes of other parameters that result in lower staining intensity can result in a comparable result as shown above.
Leica – BOND RX
Dewax at 72°C for 30 seconds; Pretreatment in Bond Epitope Retrieval Solution (ER2 – EDTA pH9) for 20 minutes at 100°C; Peroxidase blocking for 5 minutes (room temperature), MSVA-623R 1:150 for 15 minutes (room temperature), Post primary (rabbit anti mouse) for 8 minutes (room temperature), Polymer (goat anti rabbit) for 8 minutes (room temperature), mixed DAB refine for 10 minutes (room temperature), hematoxylin for 5 minutes (room temperature).
These images reflect stainings by the protocol described above. It is of note that a comparable staining result can also be obtained by different protocols. In general, a longer pretreatment, a longer incubation time of the primary antibody, a higher antibody concentration, a higher temperature during incubation, and a longer incubation time of Post primary and or the Polymer result in stronger staining, potentially at the cost of more background staining. Modifications of the protocol with a strengthening effect on staining intensity in combination with changes of other parameters that result in lower staining intensity can result in a comparable result as shown above.
Roche – Ventana Discovery ULTRA
Pretreatment for 64 minutes at 100°C (pH 8,4); CM peroxidase blocking for 12 minutes (room temperature), MSVA-623R 1:150 for 20 minutes at 36°C, secondary antibody (anti-rabbit HQ) for 12 minutes at 36°C, anti-HQ HRP for 12 minutes at room temperature, DAB at room temperature, hematoxylin II at room temperature for 8 minutes, bluing reagent at room temperature for 4 minutes.
These images depict staining results obtained by the protocol described above. It is of note, that the Ventana machines generally require higher antibody concentrations than other commonly used autostainers because the antibodies are automatically diluted during the procedure. Various other protocols can result in an identical result as shown above. A longer pretreatment, a longer incubation time of the primary antibody, a higher antibody concentration, a higher temperature during incubation, and a longer incubation time of secondary antibody and or the anti-HQ HRP result in stronger staining, potentially at the cost of more background staining.
Potential Research Applications
- Nucleolin is a promising target for anti-cancer therapy and should be further studied.
- Studying the molecular activities of cell-surface nucleolin and the distribution of nucleolin in cells is warranted.
- The interactions of surface nucleolin with ligands are poorly understood.
- Distinction of nuclei and nucleoli in multicolor immunohistochemistry.
Evidence for Antibody Specificity in IHC
There are two ways how the specificity of antibodies can be documented for immunohistochemistry on formalin fixed tissues. These are: 1. comparison with a second independent method for target expression measurement across a large number of different tissue types (orthogonal strategy), and 2. Comparison with one or several independent antibodies for the same target and showing that all positive staining results are also seen with other antibodies for the same target (independent antibody strategy).
Orthogonal validation is not suited for validation of antibodies for proteins that are expressed in every cell of every organ.
Comparison of antibodies: Specific nucleolin staining by MSVA-623R is supported by the perfect match with the expected staining pattern of a purely nuclear staining in normal tissues with nucleolar predominance. This staining pattern also matches the stainings observed by the antibody CAB004210 used in the analyses shown in the Human Protein Atlas (Tissue expression Nucleolin).