445,00 € – 2.995,00 €
Product details
Synonyms = ALPS5; CD152; Celiac disease 3 (CELIAC3); Cytotoxic T-lymphocyte-associated antigen 4 (CTLA4); GRD4; GSE; ICOS; Insulin-dependent Diabetes Mellitus 12 (IDDM12)
Antibody type = Recombinant Rabbit monoclonal / IgG
Clone = MSVA-152R
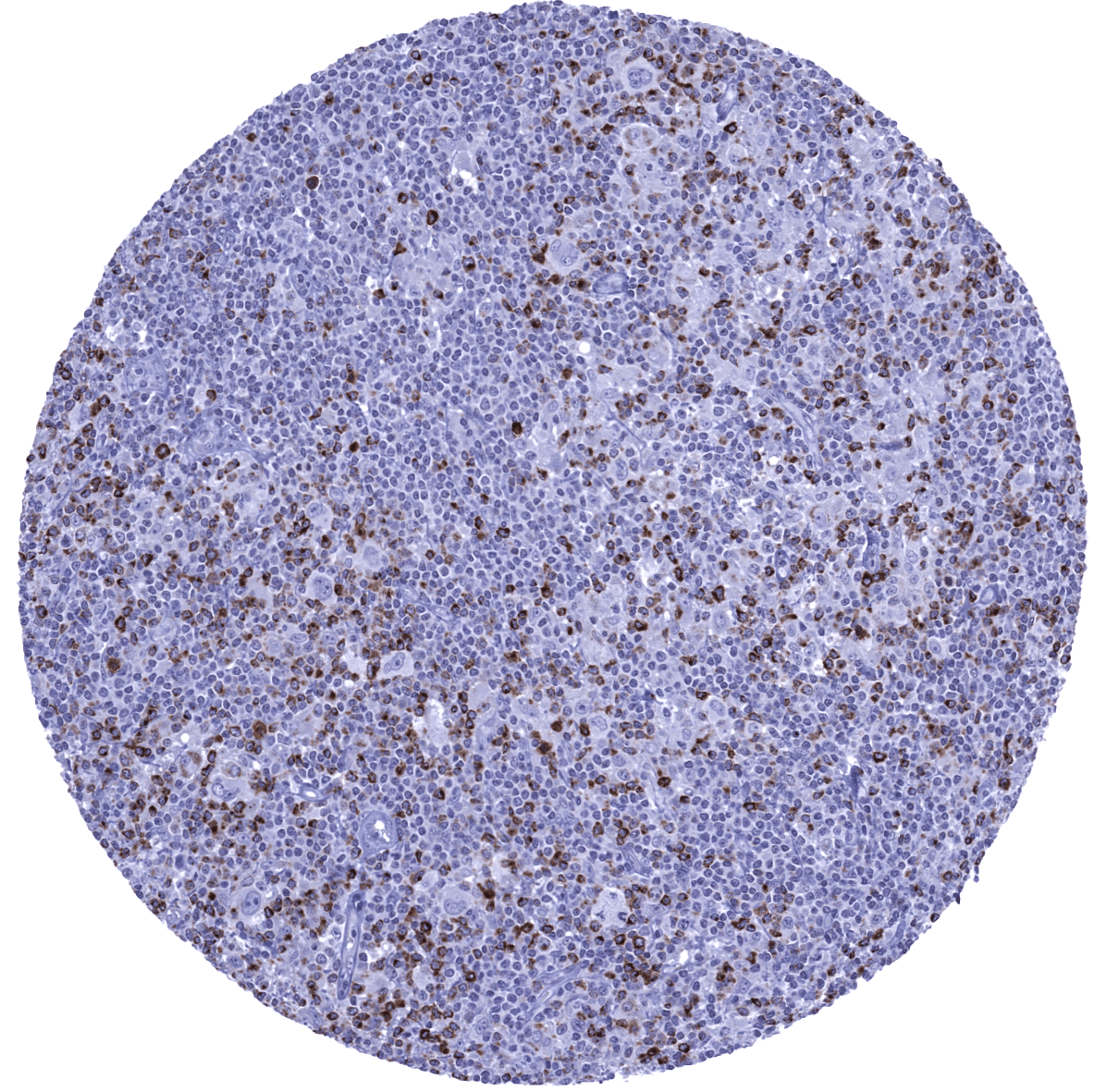
Positive control = Tonsil: A moderate strong membranous and cytoplasmic CTLA4 immunostaining should be seen in a subset of T-lymphocytes with strongest staining in follicular T-helper cells of germinal centres.
Negative control = Tonsil: The majority of lymphocytes in germinal centres and all epithelial cells should stain negative.
Cellular localization = Cell surface.
Reactivity = Human
Application = Immunohistochemistry
Dilution = 1:50-1:100
Intended Use = Research Use Only
Relevance of Antibody
CTLA-4 is a pivotal target in immune-oncology.
Biology Behind
CTLA-4 (cytotoxic T-lymphocyte-associated protein 4) is a member of the immunoglobulin superfamily. Its expression is regulated by several transcription factors including FOXP3 which determines the lineage of regulatory T-cells. CTLA-4 is thus constitutively expressed in regulatory T cells while it is upregulated in other T cells after activation only. The role of CTLA-4 in signaling depends on cellular localization of CTLA-4. In resting T cells, CTLA-4 localizes intracellularly. Upon T cell stimulation, CTLA-4 containing vesicles are exocytosed and presented on immunological synapses. CTLA4 is closely interacting with various other surface proteins such as CD28, CD80, and CD86. CTLA-4 and CD28 are co-receptors that bind to CD80 and CD86 to regulate T-cell activation. CD28 co-stimulation is required for T-cell activation, whereas CTLA-4 inhibits T-cell response by opposing the actions of CD28-mediated co-stimulation. Even though CTLA-4 is expressed by activated CD8 killer T cells, the major physiologic role of CTLA-4 appears to be through distinct effects on the two major subsets of CD4 positive T cells: down-modulation of helper T-cell activity and enhancement of regulatory T-cell suppressive activity. The CTLA-4 pathway is a commonly targeted pathway in cancer immunotherapy. CTLA-4 inhibitors for example include ipilimumab and tremelimumab.[1]
[1] Dum et al. “Semi-automated validation and quantification of CTLA-4 in 90 different tumor entities using multiple antibodies and artificial intelligence.” Laboratory Investigation January 29th 2022.
Staining Pattern in Normal Tissues
CTLA-4 staining pattern in Normal Tissues with antibody MSVA-152R (images are shown in our “Normal Tissue Gallery”)
| Brain | Cerebrum | Negative. |
| Cerebellum | Negative. | |
| Endocrine Tissues | Thyroid | CTLA-4 staining of kolloid (tolerable cross-reactivity). |
| Parathyroid | Negative. | |
| Adrenal gland | Significant granular cytoplasmic staining in adrenocortical cells (tolerable cross-reactivity). | |
| Pituitary gland | Negative. | |
| Respiratory system | Respiratory epithelium | Few lymphocytes may stain positive. |
| Lung | Few lymphocytes may stain positive. | |
| Gastrointestinal Tract | Salivary glands | Few lymphocytes may stain positive. |
| Esophagus | Few lymphocytes may stain positive. | |
| Stomach | Weak granular cytoplasmic apical CTLA-4 staining in epithelial cells (tolerable cross-reactivity). Few lymphocytes may stain positive. | |
| Colon | Weak granular cytoplasmic apical CTLA-4 staining in epithelial cells (tolerable cross-reactivity). Few lymphocytes may stain positive. | |
| Duodenum | Weak granular cytoplasmic apical CTLA-4 staining in epithelial cells (tolerable cross-reactivity). Few lymphocytes may stain positive. | |
| Rectum | Weak granular cytoplasmic apical CTLA-4 staining in epithelial cells (tolerable cross-reactivity). Few lymphocytes may stain positive. | |
| Small intestine | Weak granular cytoplasmic apical CTLA-4 staining in epithelial cells (tolerable cross-reactivity). Few lymphocytes may stain positive. | |
| Liver | Few lymphocytes may stain positive. | |
| Gallbladder | Weak granular cytoplasmic apical CTLA-4 staining in epithelial cells (tolerable cross-reactivity). Few lymphocytes may stain positive. | |
| Pancreas | Weak granular cytoplasmic CTLA-4 staining in some acinar cells (tolerable cross-reactivity). | |
| Genitourinary | Kidney | Weak granular cytoplasmic CTLA-4 staining in few tubular cells (tolerable cross-reactivity). Negative. |
| Urothelium | Few lymphocytes may stain positive. | |
| Male genital | Prostate | Few lymphocytes may stain positive. |
| Seminal vesicles | Negative. | |
| Testis | Negative. | |
| Epididymis | Weak granular cytoplasmic apical CTLA-4 staining in tall columnar cells (tolerable cross-reactivity). | |
| Female genital | Breast | Negative. |
| Uterus, ectocervix | Few lymphocytes may stain positive. | |
| Uterus endocervix | Few lymphocytes may stain positive. | |
| Uterus, endometrium | Few lymphocytes may stain positive. | |
| Fallopian Tube | Few lymphocytes may stain positive. | |
| Ovary | Negative. | |
| Placenta early | Negative. | |
| Placenta mature | Granular cytoplasmic staining in decidua cells (tolerable cross-reactivity). | |
| Amnion | Negative. | |
| Chorion | Negative. | |
| Skin | Epidermis | Few lymphocytes may stain positive |
| Sebaceous glands | Few lymphocytes may stain positive. | |
| Muscle/connective tissue | Heart muscle | Negative. |
| Skeletal muscle | Negative. | |
| Smooth muscle | Negative. | |
| Fat | Negative. | |
| Bone marrow/lymphoid | Bone marrow | Few lymphocytes may stain positive. |
| Lymph node | Moderate to strong staining in a fraction of lymphocytes showing a T-cell distribution. | |
| Spleen | Some lymphocytes show a positive staining. | |
| Thymus | Some lymphocytes show a positive staining. | |
| Tonsil | Moderate to strong staining in a fraction of lymphocytes showing a T-cell distribution. | |
| Remarks | A strong, predominantly membranous CTLA-4 staining occurs in a subset of T-lymphocytes including regulatory T-cells. Such cells can occur in all tissues.
The antibody also shows some cross-reactivities. Given the strong staining of CTLA-4 expressing lymphocytes, these are considered tolerable. |
These findings are largely consistent with the RNA and protein data described in the Human Protein Atlas (Tissue expression CTLA-4)
Positive control: Tonsil: A moderate strong membranous and cytoplasmic CTLA4 immunostaining should be seen in a subset of T-lymphocytes with strongest staining in follicular T-helper cells of germinal centres.
Negative control: Tonsil: The majority of lymphocytes in germinal centres and all epithelial cells should stain negative.
Staining Pattern in Relevant Tumor Types
CTLA-4 positive lymphocytes occur in a variable number in virtually all tumors.
The TCGA findings on CTLA-4 / CD152 RNA expression in different tumor categories have been summarized in the Human Protein Atlas.
Compatibility of Antibodies
No data available at the moment
Protocol Recommendations
IHC users have different preferences on how the stains should look like. Some prefer high staining intensity of the target stain and even accept some background. Others favor absolute specificity and lighter target stains. Factors that invariably lead to more intense staining include higher concentration of the antibody and visualization tools, longer incubation time, higher temperature during incubation, higher temperature and longer duration of the heat induced epitope retrieval (slide pretreatment). The impact of the pH during slide pretreatment has variable effects and depends on the antibody and the target protein.
All images and data shown here and in our image galleries are obtained by the manual protocol described below. Other protocols resulting in equivalent staining are described as well.
Manual protocol
Freshly cut sections should be used (less than 10 days between cutting and staining). Heat-induced antigen retrieval for 5 minutes in an autoclave at 121°C in pH 7,8 Target Retrieval Solution buffer. Apply MSVA-152R at a dilution of 1:75 at 37°C for 60 minutes. Visualization of bound antibody by the EnVision Kit (Dako, Agilent) according to the manufacturer’s directions.
Agilent / Dako – Autostainer Link 48
Pretreatment in PT-Link for 30 minutes at 95°C (pH high); FLEX peroxidase blocking for 5 minutes (room temperature), MSVA-152R 1:75 for 20 minutes (room temperature), FLEX+ mouse/rabbit (LINKER) for 15 minutes (room temperature), horseradish peroxidase (HRP) for 20 minutes (room temperature), FLEX DAB+Sub-Chromo for 10 minutes (room temperature), FLEX hematoxylin for 5 minutes (room temperature).
These images reflect stainings by the protocol described above. It is of note that a comparable staining result can also be obtained by different protocols. In general, a longer pretreatment, a longer incubation time of the primary antibody, a higher antibody concentration, and a longer incubation time of FLEX+LINKER result in stronger staining, potentially at the cost of more background staining. Modifications of the protocol with a strengthening effect on staining intensity in combination with changes of other parameters that result in lower staining intensity can result in a comparable result as shown above.
Leica – BOND RX
Dewax at 72°C for 30 seconds; Pretreatment in Bond Epitope Retrieval Solution (ER2 – EDTA pH9) for 20 minutes at 100°C; Peroxidase blocking for 5 minutes (room temperature), MSVA-152R 1:50 for 15 minutes (room temperature), Post primary (rabbit anti mouse) for 8 minutes (room temperature), Polymer (goat anti rabbit) for 8 minutes (room temperature), mixed DAB refine for 10 minutes (room temperature), hematoxylin for 5 minutes (room temperature).
These images reflect stainings by the protocol described above. It is of note that a comparable staining result can also be obtained by different protocols. In general, a longer pretreatment, a longer incubation time of the primary antibody, a higher antibody concentration, a higher temperature during incubation, and a longer incubation time of Post primary and or the Polymer result in stronger staining, potentially at the cost of more background staining. Modifications of the protocol with a strengthening effect on staining intensity in combination with changes of other parameters that result in lower staining intensity can result in a comparable result as shown above.
Roche – Ventana Discovery ULTRA
Pretreatment for 64 minutes at 100°C (pH 8,4); CM peroxidase blocking for 12 minutes (room temperature), MSVA-152R 1:50 for 20 minutes at 36°C, secondary antibody (anti-rabbit HQ) for 12 minutes at 36°C, anti-HQ HRP for 12 minutes at room temperature, DAB at room temperature, hematoxylin II at room temperature for 8 minutes, bluing reagent at room temperature for 4 minutes.
These images depict staining results obtained by the protocol described above. It is of note, that the Ventana machines generally require higher antibody concentrations than other commonly used autostainers because the antibodies are automatically diluted during the procedure. Various other protocols can result in an identical result as shown above. A longer pretreatment, a longer incubation time of the primary antibody, a higher antibody concentration, a higher temperature during incubation, and a longer incubation time of secondary antibody and or the anti-HQ HRP result in stronger staining, potentially at the cost of more background staining.
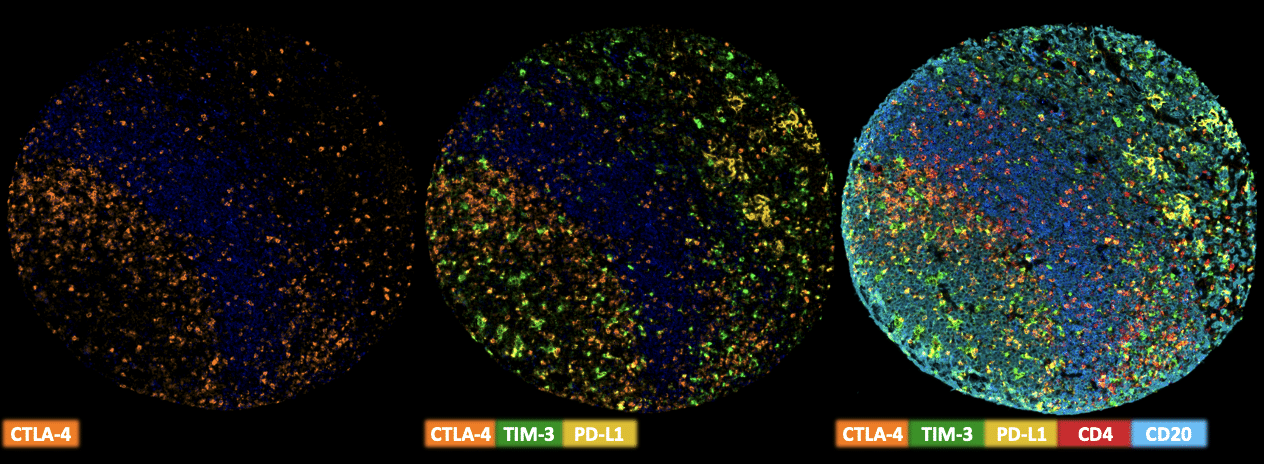
Multiplex Fluorescence Immunohistochemistry
Freshly cut sections should be used (less than 10 days between cutting and staining). Slides were initially boiled in an autoclave (30 min at 100–120 °C in pH9 buffer) for antigen retrieval. The antibody panel consists of CTLA-4 (MSVA-152R 1:75), TIM3 (MSVA-366R – 1:150), PD-L1 (MSVA-711R – 1:150), CD4 (MSVA-004R – 1:150) & CD20 (MSVA-020R – 1:150). The OPAL dye kit (Cat# NEL811001KT, AKOYA Biosciences, Menlo Park, California, United States) according to the manufacturer’s directions was used to detect the primary antibodies.
Potential Research Applications
- Quantification of CTLA-4 positive lymphocytes (The antibody is suited for multicolor immunofluorescence)
Evidence for Antibody Specificity in IHC
There are two ways, how the specificity of antibodies can be documented for immunohistochemistry on formalin fixed tissues. These are: 1. comparison with a second independent method for measuring target expression across a large number of different tissue types (orthogonal strategy), and 2. Comparison with one or several independent antibodies for the same target and showing that all positive staining results are also seen with other antibodies for the same target (independent antibody strategy).
Orthogonal validation: As a standard orthogonal validation process for MSVA antibodies, RNA data summarized in the Human Protein Atlas (Tissue expression CTLA-4) are used for comparison. However, this procedure has limited validity for proteins that are expressed in significant subsets of lymphocytes because these cells occur in virtually all organs. The validation of MSVA-152R is therefore based on a comparison with a second independent (commercial) CTLA-4 antibody (termed “Validation-antibody”). This comparison of antibodies revealed identical staining patterns for MSVA-152R and the “Validation-antibody” in lymphatic tissues with identical subsets of lymphocytes staining. In addition, both antibodies showed antibody specific cross-reactivities with different other tissues. Given the current lack of CTLA-4 antibodies without such cross-reactivities, we consider these as “tolerable”.[1]
[1] Dum et al. “Semi-automated validation and quantification of CTLA-4 in 90 different tumor entities using multiple antibodies and artificial intelligence.” Laboratory Investigation January 29th 2022.
Antibody Comparison: MSVA-152R vs another commercially available CTLA-4 antibody called “Validation Antibody”