345,00 € – 1.195,00 €
Product details
Synonyms = p185, CD340, Verb b2 Erythroblastic Leukemia Viral Oncogene Homolog 2, Neuro/Glioblastoma Derived Oncogene Homolog
Antibody type = Recombinant Rabbit monoclonal / Rabbit IgG
Clone = MSVA-340R
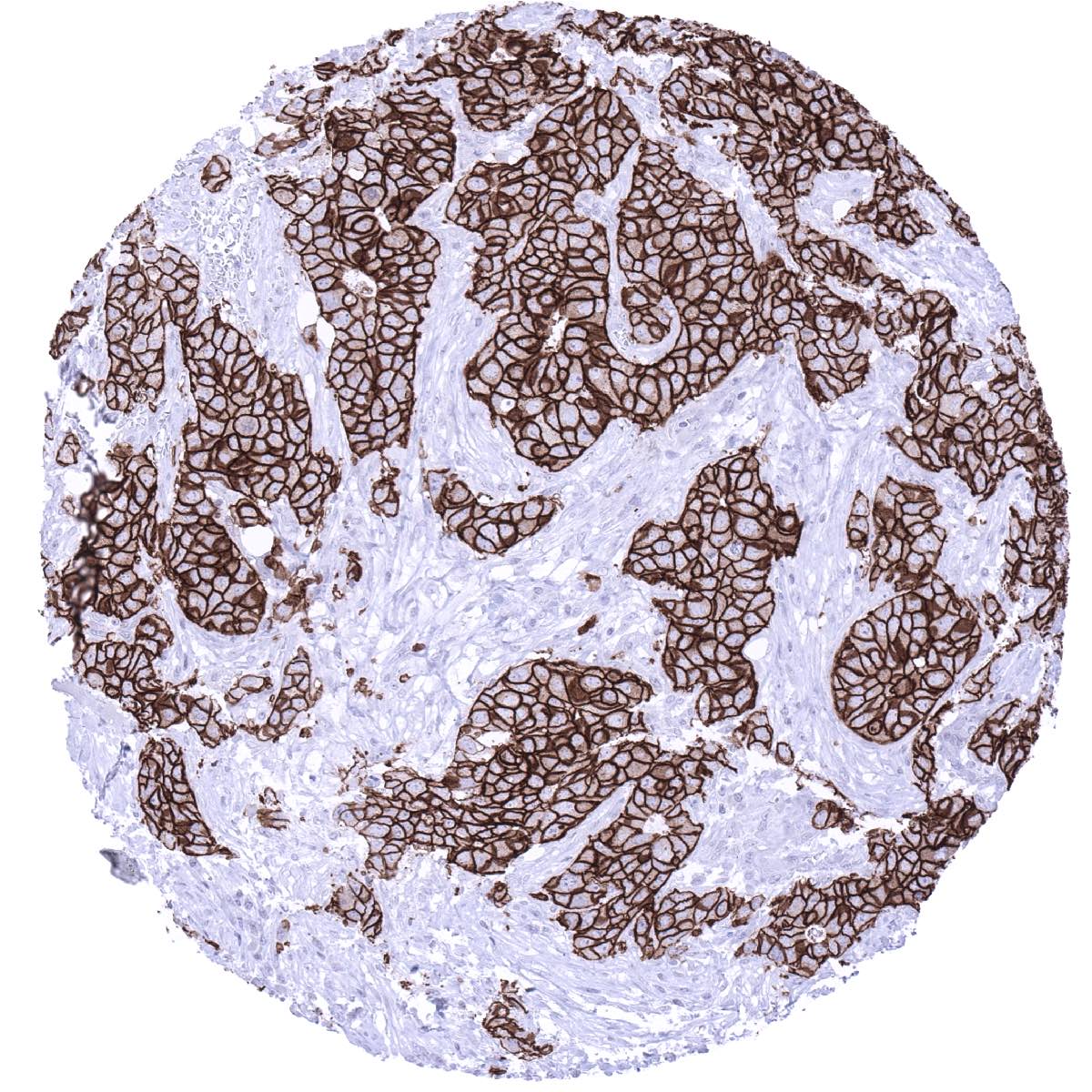
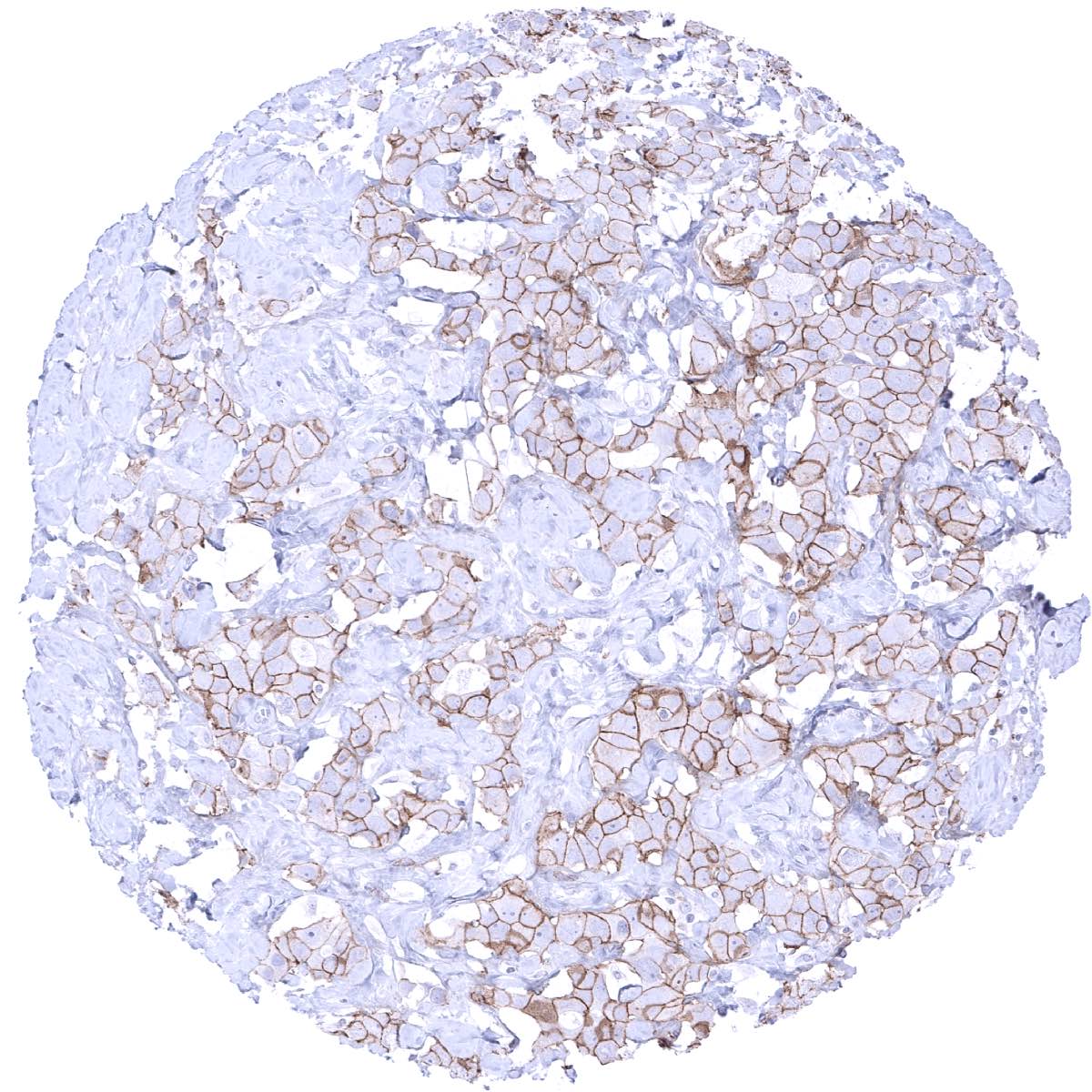
Positive control = Colon: Breast cancers with HER2 amplification should show strong positivity. Placenta: A faint staining should be seen at the surface membrane of the syncytiotrophoblast.
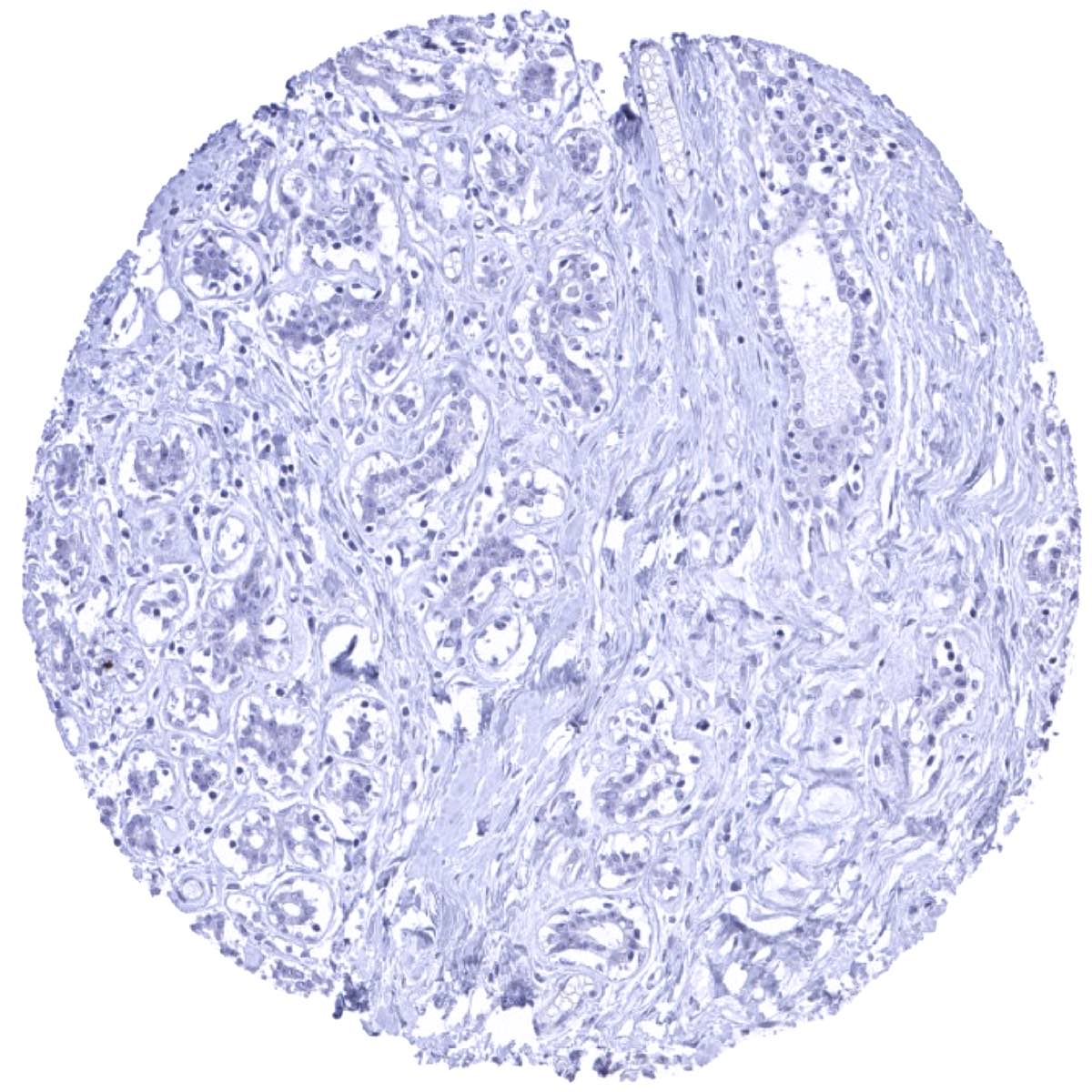
Negative control = Breast: Normal epithelial cells must not show HER2 immunostaining.
Cellular localization = Cell surface
Reactivity = Human
Application = Immunohistochemistry
Dilution = 1:50 – 1:200
Intended Use = Research Use Only
Relevance of Antibody
HER2 is an oncogenic cell surface receptor and therapeutic target.
Biology Behind
HER2, also termed erbB-2 (erythroblastic oncogene B), is a receptor tyrosine-protein kinase coded by the ERBB2 gene located at 17q21. In contrast, to other members of the ERBB-family, HER2 has no known ligand. HER2 acts through dimerization with the other three family members erbb1 (EGFR), erbb3, and erbb4. HER2 is the preferred dimerization partner of all other ErbB receptors. Dimerization results in the autophosphorylation of tyrosine residues within the cytoplasmic domain of the receptors and initiates a variety of signaling pathways. HER2 activation occurs in various tumors, mostly through gene amplification which results in high level overexpression. HER2 mutations occur less frequently but can also lead to permanent activation and dimerization in the absence of HER2 overexpression. HER2 is an ideal drug target because gene amplification leads to HER2 expression levels that are far higher than what can be observed in normal tissues. HER2 targeting drugs include trastuzumab (Herceptin), lapatinib, margetuximab, pertuzumab, and others.
Staining Pattern in Normal Tissues
HER2 expression occurs in many different tissues, but usually at a rather low level. Because HER2 immunostaining is primarily used for detection of HER2 overexpressing tumors, it is typically calibrated in a way that it results in a detectable staining in only a few normal tissues. Using such a “low sensitivity approach” a faint or weak membranous HER2 immunostaining can be seen (in some samples) at the basolateral membranes of colon apical mucosa, in luminal cells of atrophic prostate glands, basal cells of prostate glands, basal cells of seminal vesicle, and on apical membranes of the placenta.
RNA and protein expression data of HER2 findings are also described in the Human Protein Atlas (Tissue expression HER2)
Appropriate control tissues: Considering the role of HER2 testing in breast cancer, breast cancers with known HER2 amplification are highly useful as tissue controls. In appropriately calibrated HER2 tests, scores 0 or 1+ are almost never (<2%) seen in HER-2 amplified tumors, while score 3+ is linked to gene amplification in 90-100% of cases. In samples with an IHC score of 2+, less than one third of cases are usually showing HER-2 gene amplification. Inclusion of 2+ tumors with and without HER-2 gene amplification in tissue sets used for quality management is essential to assure precision and performance stability of an IHC HER-2 assay.
Suggested positive tissue control: Breast cancers with HER2 amplification should show strong positivity. Placenta: A faint staining should be seen at the surface membrane of the syncytiotrophoblast.
Suggested negative tissue control: Breast: Normal epithelial cells must not show HER2 immunostaining.
Staining Pattern in Relevant Tumor Types
HER2 overexpression and HER2 amplification is particularly common in breast and gastric cancer but can – at lower frequency – also occur in many other tumor entities.
The TCGA findings on HER2 RNA expression in different tumor categories have been summarized in the Human Protein Atlas.
Protocol Recommendations
IHC users have different preferences on how the stains should look like. Some prefer high staining intensity of the target stain and even accept some background. Others favor absolute specificity and lighter target stains. Factors that invariably lead to more intense staining include higher concentration of the antibody and visualization tools, longer incubation time, higher temperature during incubation, higher temperature and longer duration of the heat induced epitope retrieval (slide pretreatment). The impact of the pH during slide pretreatment has variable effects and depends on the antibody and the target protein.
All images and data shown here and in our image galleries are obtained by the manual protocol described below. Other protocols resulting in equivalent staining are described as well.
Manual protocol
Freshly cut sections should be used (less than 10 days between cutting and staining). Heat-induced antigen retrieval for 5 minutes in an autoclave at 121°C in pH 7,8 Target Retrieval Solution buffer. Apply MSVA-340R at a dilution of 1:50 at 37°C for 60 minutes. Visualization of bound antibody by the EnVision Kit (Dako, Agilent) according to the manufacturer’s directions.
Agilent / Dako – Autostainer Link 48
Pretreatment in PT-Link for 30 minutes at 95°C (pH high); FLEX peroxidase blocking for 5 minutes (room temperature), MSVA-340R 1:200 for 20 minutes (room temperature), FLEX+ mouse/rabbit (LINKER) for 15 minutes (room temperature), horseradish peroxidase (HRP) for 20 minutes (room temperature), FLEX DAB+Sub-Chromo for 10 minutes (room temperature), FLEX hematoxylin for 5 minutes (room temperature).
These images reflect stainings by the protocol described above. It is of note that a comparable staining result can also be obtained by different protocols. In general, a longer pretreatment, a longer incubation time of the primary antibody, a higher antibody concentration, and a longer incubation time of FLEX+LINKER result in stronger staining, potentially at the cost of more background staining. Modifications of the protocol with a strengthening effect on staining intensity in combination with changes of other parameters that result in lower staining intensity can result in a comparable result as shown above.
Leica – BOND RX
Dewax at 72°C for 30 seconds; Pretreatment in Bond Epitope Retrieval Solution (ER2 – EDTA pH9) for 20 minutes at 100°C; Peroxidase blocking for 5 minutes (room temperature), MSVA-MSVA-340R 1:200 for 15 minutes (room temperature), Post primary (rabbit anti mouse) for 8 minutes (room temperature), Polymer (goat anti rabbit) for 8 minutes (room temperature), mixed DAB refine for 10 minutes (room temperature), hematoxylin for 5 minutes (room temperature).
These images reflect stainings by the protocol described above. It is of note that a comparable staining result can also be obtained by different protocols. In general, a longer pretreatment, a longer incubation time of the primary antibody, a higher antibody concentration, a higher temperature during incubation, and a longer incubation time of Post primary and or the Polymer result in stronger staining, potentially at the cost of more background staining. Modifications of the protocol with a strengthening effect on staining intensity in combination with changes of other parameters that result in lower staining intensity can result in a comparable result as shown above.
Roche – Ventana Discovery ULTRA
Pretreatment for 64 minutes at 100°C (pH 8,4); CM peroxidase blocking for 12 minutes (room temperature), MSVA-340R 1:100 for 20 minutes at 36°C, secondary antibody (anti-rabbit HQ) for 12 minutes at 36°C, anti-HQ HRP for 12 minutes at room temperature, DAB at room temperature, hematoxylin II at room temperature for 8 minutes, bluing reagent at room temperature for 4 minutes.
These images depict staining results obtained by the protocol described above. It is of note, that the Ventana machines generally require higher antibody concentrations than other commonly used autostainers because the antibodies are automatically diluted during the procedure. Various other protocols can result in an identical result as shown above. A longer pretreatment, a longer incubation time of the primary antibody, a higher antibody concentration, a higher temperature during incubation, and a longer incubation time of secondary antibody and or the anti-HQ HRP result in stronger staining, potentially at the cost of more background staining.
Potential Research Applications
- The prevalence and extent of HER2 heterogeneity is unknown for most cancer types.
- New anti-HER2 drugs are under development.
- About 60% of HER2 positive cancers develop resistance to trastuzumab. Biomarkers are needed to predict trastuzumab resistance.
Evidence for Antibody Specificity in IHC
There are two ways how the specificity of antibodies can be documented for immunohistochemistry on formalin fixed tissues. These are: 1. Comparison with a second independent method for target expression measurement across a large number of different tissue types (orthogonal strategy), and 2. Comparison with one or several independent antibodies for the same target and showing that all positive staining results are also seen with other antibodies for the same target (independent antibody strategy).
Orthogonal validation: Orthogonal validation is not well suited for HER2 antibodies because very low level HER2 RNA expression has been described in many organs. However, within these organs, HER2 expression is limited to specific cell types and varies more between cell types than between organs. Available RNA databases identified comparable levels of HER2 RNA expression in many organs Human Protein Atlas (Tissue expression HER2) which makes comparison of IHC findings with RNA results difficult.
Comparison of antibodies: For the antibody MSVA-340R specificity for HER2 is suggested by the strong concordance of its immunostaining data with data obtained by an independent second commercially available FDA approved antibody (termed “validation antibody”).