145,00 € – 595,00 €
Product details
Synonyms = Carcinoembryonic Antigen-related Cell Adhesion Molecule 5, CEACAM5, CD66, Biliary Glycoprotein (BGP-1)
Antibody type = Recombinant Rabbit monoclonal / IgG
Clone = MSVA-465R
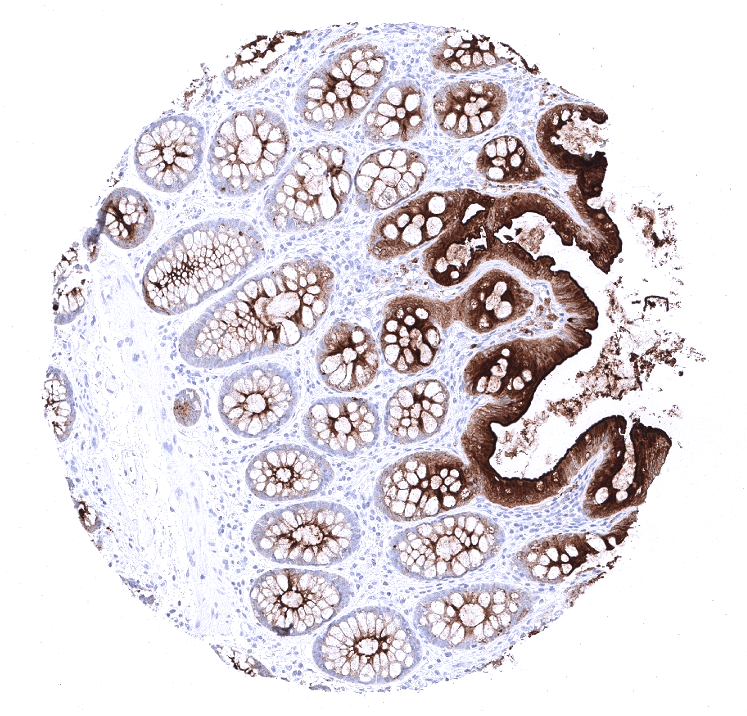
Positive control = Colon: An at least moderate cytoplasmic CEA staining should be seen in the vast majority of columnar epithelial cells.
Negative control = Colon: CEA staining should not be seen in stromal cells or smooth muscle.
Cellular localization = Cytoplasmic and luminal surface
Reactivity = Human
Application = Immunohistochemistry
Dilution = 1:100 – 1:200
Intended Use = Research Use Only
Relevance of Antibody
CEA is a protein with variable expression in epithelial tumor types.
Biology Behind
Carcinoembryonic antigen (CEA; CEACAM5, CD66e) is a 180-kDA cell surface glycoprotein involved in cell adhesion that is extensively produced during fetal development but limited to few normal tissues already before birth. It belongs to a family of more than 30 closely related CEA like proteins. CEA is clinically important because it is overexpressed in various cancers. Since tumoral CEA is also shed into the bloodstream, serum CEA measurement can be utilized for early detection and recurrence monitoring of cancer. Because the frequency of CEA expression varies between tumor types, CEA immunohistochemistry is regularly employed in surgical pathology for cancer classification such as for the difficult distinction of pleural mesothelioma from adenocarcinoma. Moreover, CEA is also a potential drug target. For example, cibisatamab, a bispecific antibody with binding sites for CD3 and CEA showed antitumor activity with radiological shrinkage in patients treated with cibisatamab monotherapy and in with cibisatamab and PD-L1-inhibiting antibodies in a phase I trial.
Staining Pattern in Normal Tissues
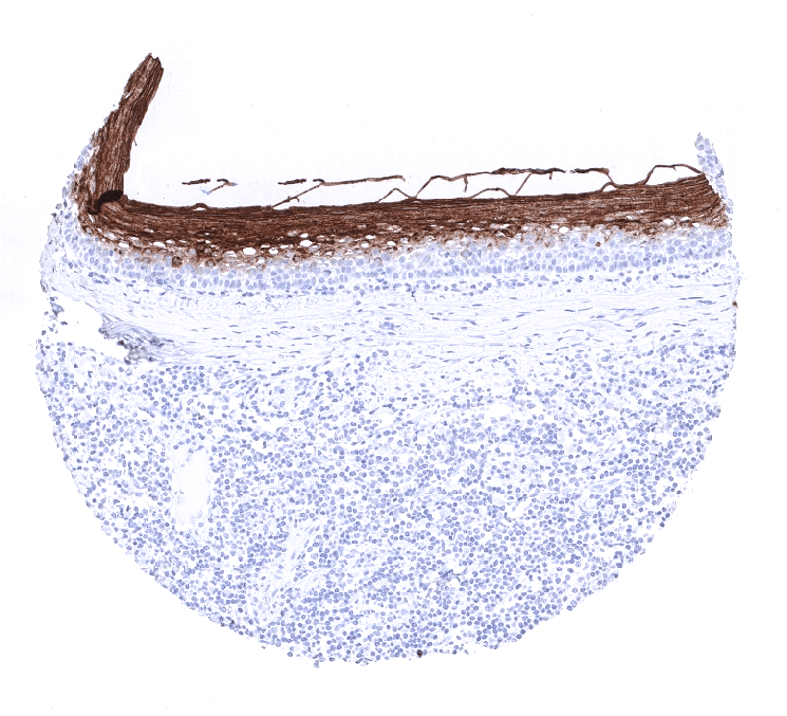
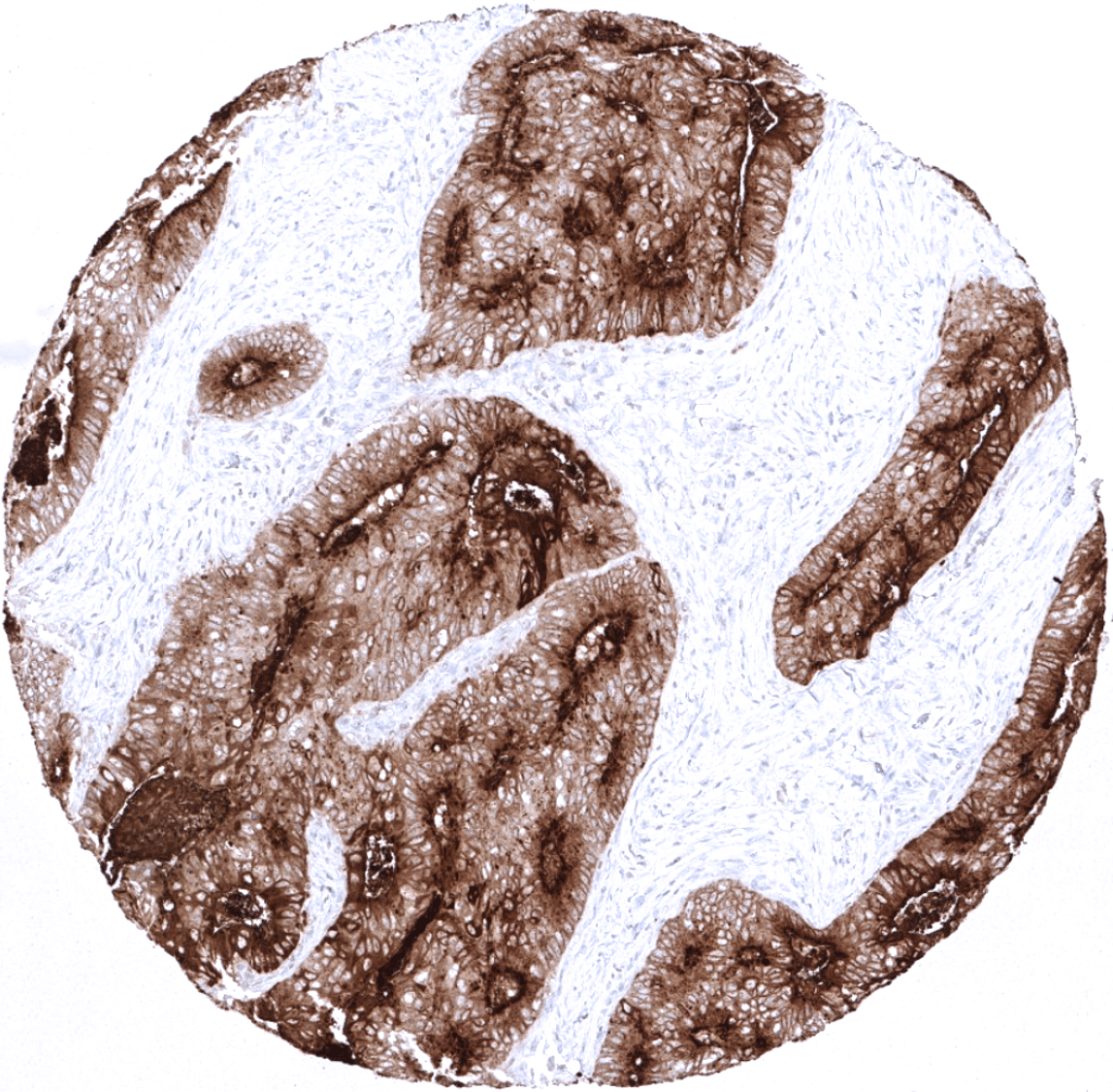
Moderate to strong CEA expression is seen in the upper layers of non-keratinizing squamous epithelium of oral cavity, lip, tonsil surface and crypts, and ectocervix while all cell layers stain in the esophagus. Strong expression occurs in transitional epithelium of the anal canal and in most cells of Hassal’s corpuscles in the thymus. Stomach mucosa surface cell layers show moderate to strong CEA positivity, while glands are negative or much less stained. Duodenum and small intestine show a moderate intensity staining of goblet cells primarily of surface epithelium while deeper glands are mostly negative. Strongest CEA staining in the GIT is seen in the colorectal area including appendix. Here the staining is again always strongest in the surface cells and upper cell layers of crypts. Salivary glands show a weak membranous apical staining of mucinous cells. Weak to moderate staining occurs in goblet cells of respiratory epithelium from bronchus and paranasal sinus. Staining can be seen in few pneumocytes. The urothelium is usually CEA negative but occasional weak positivity occurs in umbrella cells. CEA staining is absent in mesenchymal structures, lymphatic and hematopoietic cells, skin, liver, kidney, gall bladder epithelium, Brunner glands, prostate, seminal vesicle, epididymis, testis, breast, endocervix, endometrium, ovary including corpus luteum and follicular cysts, placenta, adrenal gland, thyroid, cerebrum, cerebellum, adeno- and neurohypophysis.
These findings are largely comparable to the RNA and protein data described in the Human Protein Atlas (Tissue expression CEA).
Suggested positive tissue control: Colon or appendix: An at least moderate cytoplasmic staining should be seen in the vast majority of columnar epithelial cells.
Suggested negative tissue control: Colon or appendix: Staining should not be seen in stroma cells or smooth muscle.
Staining Pattern in Relevant Tumor Types
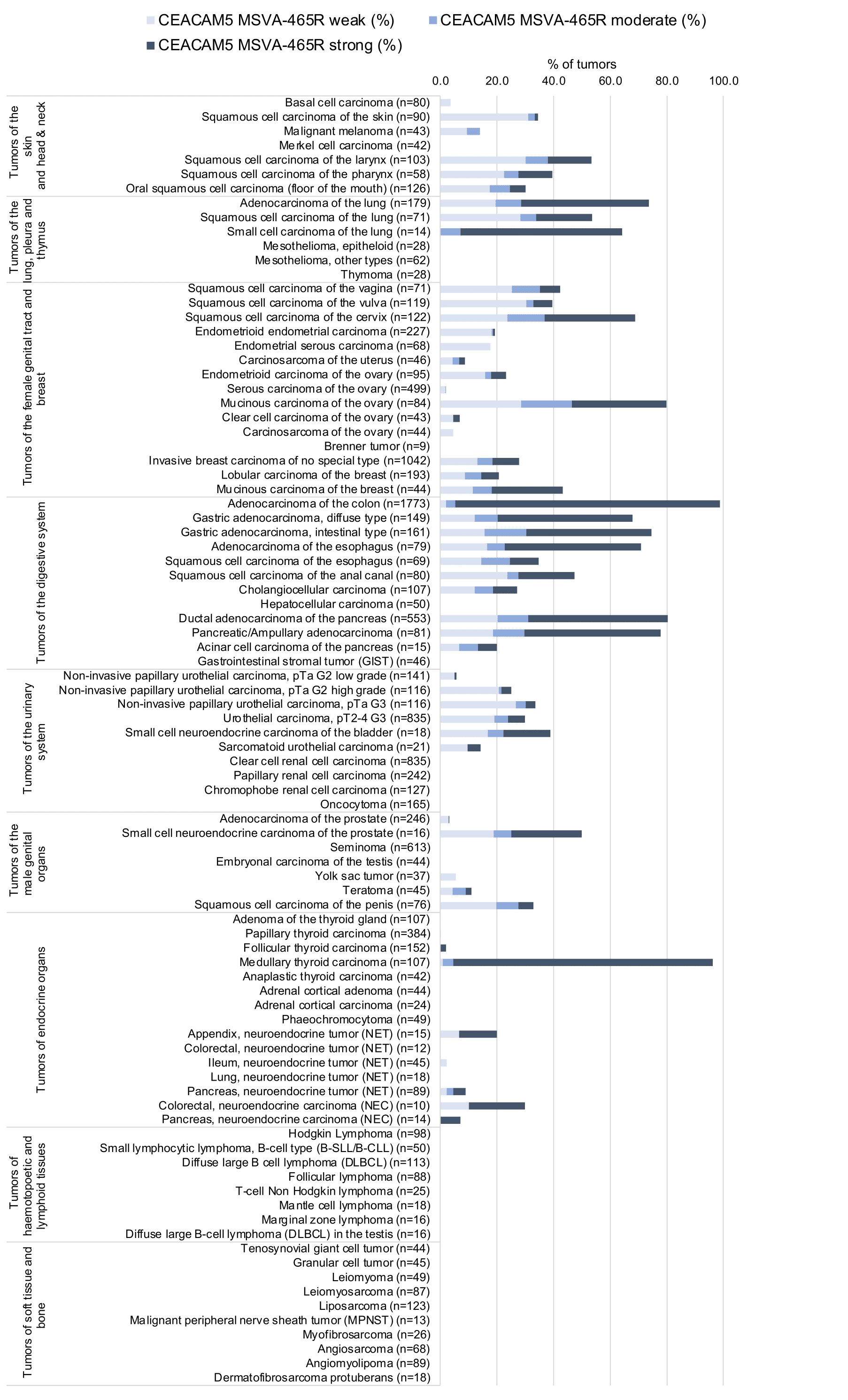
CEA is expressed in almost all colorectal adenocarcinomas and medullary carcinomas of the thyroid. CEA expression is also high in a large fraction of adenocarcinomas of the salivary glands, esophagus, stomach, biliary tract, pancreas, lung, uterine cervix and ovary (mucinous carcinomas). CEA is less frequently seen in squamous cell carcinomas, breast cancer, urothelial cancer, endometrioid carcinoma, and neuroendocrine tumors. Tumor types that seldom express CEA include: ovarian serous and clear cell carcinoma, renal cell carcinoma, adrenal cortical carcinoma, germ cell tumors of the testis, prostate adenocarcinoma, hepatocellular carcinoma, and thyroid carcinoma (except medullary).
The TCGA findings on CEA RNA expression in different tumor categories have been summarized in the Human Protein Atlas.
Compatibility of Antibodies
CEA (MSVA-465R) publication summary
Relevant publication: Jansen et al.: “Carcinoembryonic Antigen Expression in Human Tumors: A Tissue Microarray Study on 13,725 Tumors” Published in Cancers (Basel). 2024 Dec 3;16(23):4052. PMID: 39682238
A total of 13’725 tumors from 120 different tumor categories were successfully analyzed for CEA by using the following protocol: Heat-induced antigen retrieval for 5 minutes in an autoclave at 121°C in pH 9 Target Retrieval Solution buffer. MSVA-465R at a dilution of 1:150 at 37°C for 60 minutes. Visualization of bound antibody by the EnVision Kit (Dako, Agilent). This protocol was also used for all CEA immunostainings depicted in our tumor and normal tissue galleries.
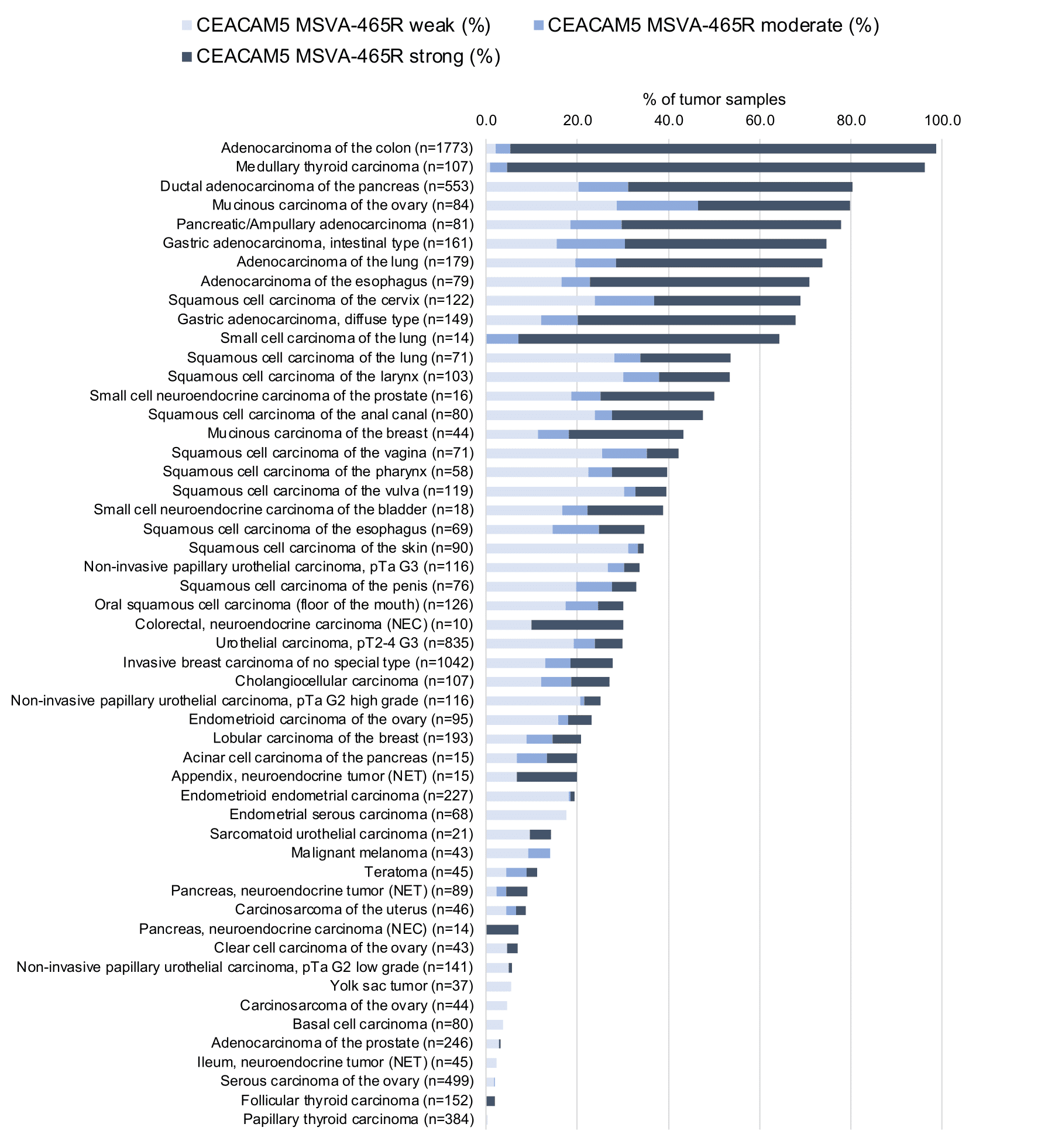
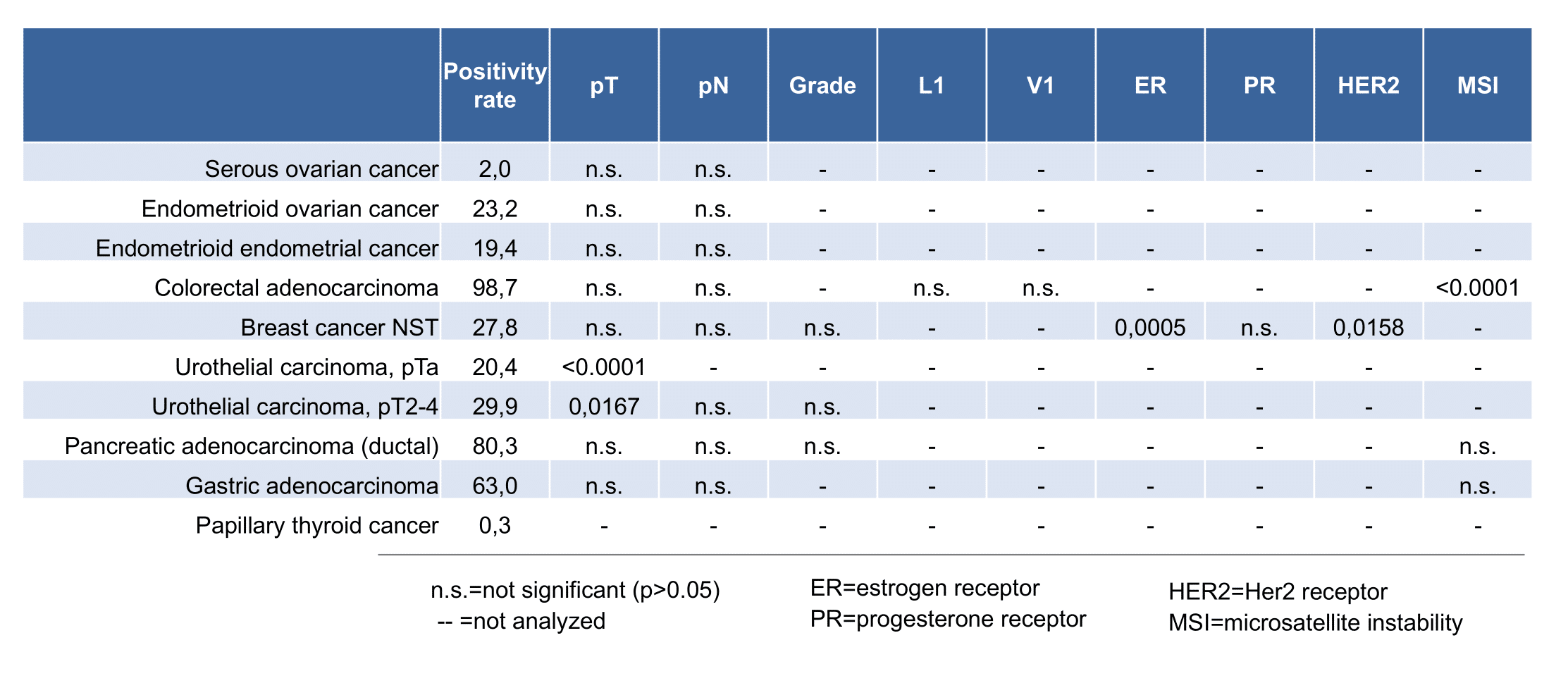
At least one case with a positive CEA immunostaining was seen in 65 and at least one case with a strong CEA immunostaining occurred in 29 of 120 tumor categories. The distribution of positive staining results is shown in an “organ-systematic” (Figure 1) and in a “ranking order” figure (Figure 2) below (images based on data from Jansen et al.). Data on associations with histopathological and clinical parameters of tumor aggressiveness in several cancer types are also summarized below (Figure 3; based on data described by Jansen et al.).
Authors conclusions on diagnostic utility with respect to the distinction of benign versus malignant (Jansen et al.):
- not applicable
Authors conclusions on diagnostic utility with respect to the distinction of different tumor entities (Jansen et al.):
- Distinction of malignant mesothelioma (mostly negative; 0% of 90) from pulmonary adenocarcinoma (mostly positive; 74% of 179).
- Distinction of hepatocellular carcinoma (mostly negative; 0% of 50) from metastases to the liver (often positive).
Authors conclusions on prognostic/predictive role of CEA expression (Jansen et al.):
- High CEA expression was associated with advanced pT stage (p < 0.0001) and muscle-invasive growth (p < 0.0001) in urothelial carcinoma of the urinary bladder.
- The absence of clear-cut associations with histopathological parameters of tumor aggressiveness in several other tumor entities and the lack of a prognostic significance in invasive breast cancers of no special type was viewed by the authors as an argument against a strong and clinically relevant prognostic role of CEA expression in cancer.
Figure 1. CEA staining in cancer (“organ-systematic”; according to Jansen et al.)
Figure 2. CEA staining in cancer (“ranking list”; according to Jansen et al.)
Figure 3. CEA staining and tumor phenotype (according to Jansen et al..)
Protocol Recommendations
IHC users have different preferences on how the stains should look like. Some prefer high staining intensity of the target stain and even accept some background. Others favor absolute specificity and lighter target stains. Factors that invariably lead to more intense staining include higher concentration of the antibody and visualization tools, longer incubation time, higher temperature during incubation, higher temperature and longer duration of the heat induced epitope retrieval (slide pretreatment). The impact of the pH during slide pretreatment has variable effects and depends on the antibody and the target protein. Accordingly, multiple different protocols can generate identical staining results.
All images and data shown here and in our image galleries are obtained by the manual protocol described below. Other protocols resulting in equivalent staining are described as well.
Manual protocol
Freshly cut sections should be used (less than 10 days between cutting and staining). Heat-induced antigen retrieval for 5 minutes in an autoclave at 121°C in pH9 Target Retrieval Solution buffer. Apply MSVA-465R at a dilution of 1:150 at 37°C for 60 minutes. Visualization of bound antibody by the EnVision Kit (Dako, Agilent) according to the manufacturer’s directions.
Agilent / Dako – Autostainer Link 48
Pretreatment in PT-Link for 30 minutes at 95°C (pH high); FLEX peroxidase blocking for 5 minutes (room temperature), MSVA-465R 1:150 for 20 minutes (room temperature), FLEX+ mouse/rabbit (LINKER) for 15 minutes (room temperature), horseradish peroxidase (HRP) for 20 minutes (room temperature), FLEX DAB+Sub-Chromo for 10 minutes (room temperature), FLEX hematoxylin for 5 minutes (room temperature).
These images reflect stainings by the protocol described above. It is of note that a comparable staining result can also be obtained by different protocols. In general, a longer pretreatment, a longer incubation time of the primary antibody, a higher antibody concentration, and a longer incubation time of FLEX+LINKER result in stronger staining, potentially at the cost of more background staining. Modifications of the protocol with a strengthening effect on staining intensity in combination with changes of other parameters that result in lower staining intensity can result in a comparable result as shown above.
Leica – BOND RX
Dewax at 72°C for 30 seconds; Pretreatment in Bond Epitope Retrieval Solution (ER2 – EDTA pH9) for 20 minutes at 100°C; Peroxidase blocking for 5 minutes (room temperature), MSVA-465R 1:150 for 15 minutes (room temperature), Post primary (rabbit anti mouse) for 8 minutes (room temperature), Polymer (goat anti rabbit) for 8 minutes (room temperature), mixed DAB refine for 10 minutes (room temperature), hematoxylin for 5 minutes (room temperature).
These images reflect stainings by the protocol described above. It is of note that a comparable staining result can also be obtained by different protocols. In general, a longer pretreatment, a longer incubation time of the primary antibody, a higher antibody concentration, a higher temperature during incubation, and a longer incubation time of Post primary and or the Polymer result in stronger staining, potentially at the cost of more background staining. Modifications of the protocol with a strengthening effect on staining intensity in combination with changes of other parameters that result in lower staining intensity can result in a comparable result as shown above.
Roche – Ventana Discovery ULTRA
Pretreatment for 64 minutes at 100°C (pH 8,4); CM peroxidase blocking for 12 minutes (room temperature), MSVA-465R 1:75 for 20 minutes at 36°C, secondary antibody (anti-rabbit HQ) for 12 minutes at 36°C, anti-HQ HRP for 12 minutes at room temperature, DAB at room temperature, hematoxylin II at room temperature for 8 minutes, bluing reagent at room temperature for 4 minutes.
These images depict staining results obtained by the protocol described above. It is of note, that the Ventana machines generally require higher antibody concentrations than other commonly used autostainers because the antibodies are automatically diluted during the procedure. Various other protocols can result in an identical result as shown above. A longer pretreatment, a longer incubation time of the primary antibody, a higher antibody concentration, a higher temperature during incubation, and a longer incubation time of secondary antibody and or the anti-HQ HRP result in stronger staining, potentially at the cost of more background staining.
Impact of pH
Potential Research Applications
- The diagnostic utility of CEA expression should be evaluated in large studies comparing different cancer entities.
- The prognostic role of CEA expression is not clear for many cancers.
- The utility of CEA as a drug target is under investigation.
Evidence for Antibody Specificity in IHC
Specificity of MSVA-465R is documented by strong concordance of the data described here and the RNA data summarized in the protein atlas. In agreement with the performance of MSVA-465R, RNA expression is markedly higher in the esophagus than in other tissues covered by squamous epithelium, and also higher in the colorectum than in the stomach, duodenum and small intestine. Other findings such as the apical membrane staining of mucinous glands in agreement with data obtained by the protein atlas antibody CAB000022. In addition CEA immunostaining by MSVA-465R is absent in tissues known to not express CEA including hepatocytes, lymphocytes and mesenchymal tissues.