295,00 € – 995,00 €
Product details
Synonyms = Endoplasmic reticulum interferon stimulator; ERIS; hMITA; hSTING; Mediator of IRF3 activation; MITA; NET23; MPYS; Mitochondrial mediator of IRF3 activation; N terminal methionine proline tyrosine serine plasma membrane tetraspanner; Stimulator of interferon genes protein; Transmembrane protein 173
Antibody type = Mouse monoclonal / IgG
Clone = MSVA-515M
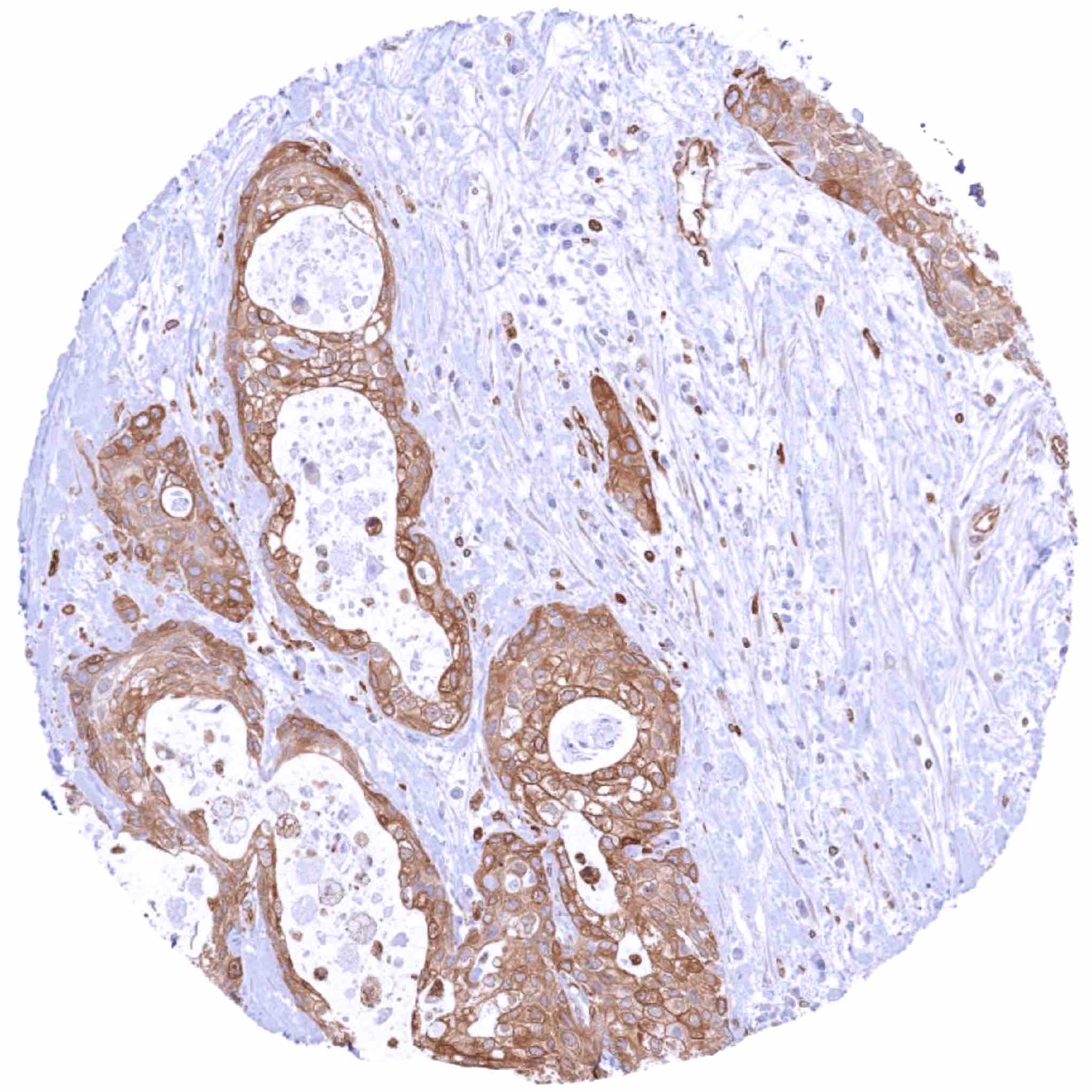
Positive control = Adrenal gland: Endothelial cells and macrophages should show a moderate to strong STING positivity.
Negative control = Adrenal gland: Adrenocortical and medullary cells should not show STING staining (Note: in case of tissue damage aberrant STING staining may occur).
Cellular localization = Cytoplasm
Reactivity = Human
Application = Immunohistochemistry
Dilution = 1:100 – 1:200
Intended Use = Research Use Only
Relevance of Antibody
STING1 is pivotal protein for innate immunity acting as a pathogen receptor.
Biology Behind
Stimulator of interferon genes (STING), also known as transmembrane protein 173 (TMEM173) is coded by the STING1 gene on chromosome 5q31.2. STING is a pathogen recognition receptor located in the endoplasmic reticulum. It is activated by cyclic dinucleotides (CDNs) which are either derived from microorganisms or synthesized by the enzyme cyclic GMP-AMP synthase (cGAS) in response to binding either host- or pathogen-derived cytosolic double-stranded (ds)DNA. STING plays an important role in innate immunity. STING induces type I interferon production when cells are infected with intracellular pathogens, such as viruses, mycobacteria and intracellular parasites. Type I interferon, mediated by STING, protects infected cells and adjacent cells from infection by autocrine and paracrine signaling. STING works as both a direct cytosolic DNA sensor (CDS) and an adaptor protein in Type I interferon signaling through different molecular mechanisms. STING activates downstream transcription factors STAT6 and IRF3 through TBK1, which are responsible for antiviral response and innate immune response against intracellular pathogens. In cancer, the cGAS/STING pathway is relevant for both tumor cells as well as neighboring immune and non-immune cells. In mice, STING deficiency facilitates development of several types of tumors while STING stimulation favors antitumor immunity. Cancer cell cGAS recognizing cytosolic DNA produces cGAMP and induces STING-dependent type-I IFN secretion. Acute STING activation is likely to exhibit type I IFN-mediated anti-tumor effect associated with cellular senescence and T lymphocyte-dependent immunity. In more advanced stages, chromosomally unstable tumors may become tolerant to chronic cGAS-STING signaling.
Staining Pattern in Normal Tissues
STING staining pattern in Normal Tissues with antibody MSVA-515M (Images shown in our “Normal Tissue Gallery”)
| Brain | Cerebrum | STING staining of endothelial cells. |
| Cerebellum | STING staining of endothelial cells. | |
| Endocrine Tissues | Thyroid | STING staining of endothelial cells. |
| Parathyroid | STING staining of endothelial cells. | |
| Adrenal gland | STING staining of endothelial cells and of macrophages. | |
| Pituitary gland | STING staining of endothelial cells and (in some samples) of a subset of epithelial cells. | |
| Respiratory system | Respiratory epithelium | Diffuse strong staining of epithelial cells. |
| Lung | Strong STING staining of macrophages. | |
| Gastrointestinal Tract | Salivary glands | STING staining of endothelial cells and of basal cells in excretory ducts. |
| Esophagus | Weak to moderate STING staining of the basal cell layers of the squamous epithelium. | |
| Stomach | Variable STING staining of epithelial cells ranging from complete lack of staining to substantial staining of most or all cells. | |
| Duodenum | Variable STING staining of epithelial cells ranging from complete lack of staining to substantial staining of most or all cells. | |
| Small intestine | Variable STING staining of epithelial cells ranging from complete lack of staining to substantial staining of most or all cells. | |
| Appendix | Variable STING staining of epithelial cells ranging from complete lack of staining to substantial staining of most or all cells. | |
| Colon | Variable STING staining of epithelial cells ranging from complete lack of staining to substantial staining of most or all cells. | |
| Rectum | Variable STING staining of epithelial cells ranging from complete lack of staining to substantial staining of most or all cells. | |
| Liver | STING staining of endothelial cells, sinus cells and inflammatory cells. | |
| Gallbladder | Variable STING staining of epithelial cells ranging from complete lack of staining to substantial staining of most or all cells. | |
| Pancreas | STING staining of endothelial cells, intercalated ducts and excretory ducts. | |
| Genitourinary | Kidney | STING staining of endothelial cells including glomeruli. |
| Urothelium | Variable STING staining ranging from weak positivity of basal cells only to strong positivity of all cell layers. | |
| Male genital | Prostate | Moderate to strong STING staining of basal cells while only few acinar cells may show some positivity. |
| Seminal vesicles | Moderate to strong STING staining of a scattered fraction of luminal epithelial and basal cells. | |
| Testis | STING staining of endothelial cells and of Leydig cells. | |
| Epididymis | Moderate to strong STING staining of basal cells in the caput and of a fraction of epithelial cells in the cauda epididymis. | |
| Female genital | Breast | STING staining of some myoepithelial cells and (weakly) of luminal cells in some samples. |
| Uterus, myometrium | STING staining of endothelials cells. | |
| Uterus, ectocervix | Weak STING staining of the basal cell layer of the squamous epithelium. | |
| Uterus endocervix | Significant STING staining of epithelial cells. | |
| Uterus, endometrium | Highly variable STING staining of epithelial cells ranging from negative to strongly positive and also of stromal cells. | |
| Fallopian Tube | Strong STING staining of epithelial cells. | |
| Ovary | A weak to moderate STING staining is regularly seen in stroma cells. | |
| Placenta early | Moderate STING staining of macrophages. | |
| Placenta mature | Moderate STING staining of macrophages. | |
| Amnion | Negative. | |
| Chorion | Negative. | |
| Skin | Epidermis | A moderate STING staining can be seen in the basal cell layer in some samples. |
| Sebaceous glands | Negative. | |
| Muscle/connective tissue | Heart muscle | STING staining of endothelials cells of small capillaries. |
| Skeletal muscle | STING staining of endothelials cells of capillaries. | |
| Smooth muscle | Mostly negative but staining of groups of smooth muscle fibres can occur in some samples. | |
| Vessel walls | STING staining of endothelials cells and of a subset of mesenchymal cells (in the aorta). | |
| Fat | Negative. | |
| Stroma | Some stromal cells may stain. | |
| Endothelium | STING staining of endothelials cells is always seen. | |
| Bone marrow/lymphoid tissue | Bone marrow | STING staining of endothelials cells and of a subset of hematopoetic cells. |
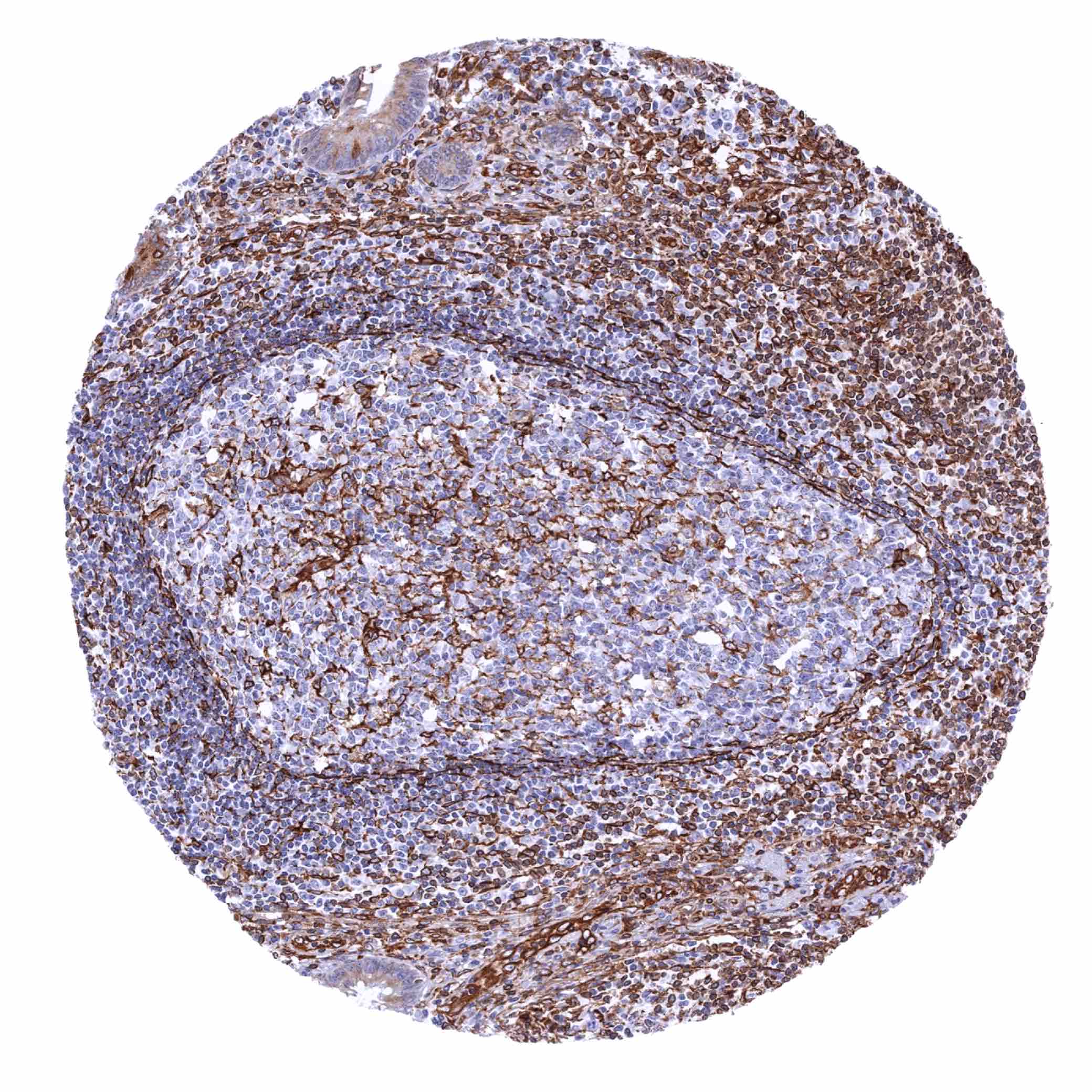
| Lymph node | Moderate to strong STING staining of a significant fraction of lymphocytes, mainly in the interfollicular zone. | |
| Spleen | Strong STING staining of endothelial cells and a significant fraction of inflammatory cells. | |
| Thymus | Moderate to strong STING staining of a significant fraction of lymphocytes in the medulla but hardly in the cortex. | |
| Tonsil | Moderate to strong STING staining of a fraction of lymphocytes, endothelial cells and of most cells of crypt and surface epithelium. | |
| Remarks | STING staining can occasionally occur in most cell types, especially in case of some morphological aspects of tissue damage. |
These findings are largely consistent with the RNA and protein data described in the Human Protein Atlas (Tissue expression STING1) All organs with documented Upk1b RNA expression (urinary bladder, kidney, prostate, gallbladder, stomach, placenta, fallopian tube, uterine cervix, tonsil) with the only exception of smooth muscle are IHC positive for MSV-515M. Given the immediate vicinity of smooth muscle to multiple Upk1b positive tissues, smooth muscle RNA positivity may represent a contamination artifact.
Positive control = Adrenal gland: Endothelial cells and macrophages should show a moderate to strong STING positivity.
Negative control = Adrenal gland: Adrenocortical and medullary cells should not show STING staining (Note: in case of tissue damage aberrant STING staining may occur).
Staining Pattern in Relevant Tumor Types
A variable STING staining of tumor cells and of stroma cells can occur in many different tumor entities.
The TCGA findings on STING1 RNA expression in different tumor categories have been summarized in the Human Protein Atlas.
Compatibility of Antibodies
STING (MSVA-515M) publication summary:
Relevant publication: Menz et al.: “Stimulator of Interferon Genes Protein (STING) Expression in Cancer Cells: A Tissue Microarray Study Evaluating More than 18,000 Tumors from 139 Different Tumor Entities” Published in Cancers (Basel) 2024, Jun 30 PMID: 39001487
A total of 15’345 tumors were successfully analyzed from 139 different tumor categories by using the following protocol: Heat-induced antigen retrieval for 5 minutes in an autoclave at 121°C in pH7,8 Target Retrieval Solution buffer. Apply MSVA-515M at a dilution of 1:150 at 37°C for 60 minutes. Visualization of bound antibody by the EnVision Kit (Dako, Agilent). This protocol was also used for all stainings depicted in our tumor and normal tissue galleries.
Almost all previous studies on STING expression in cancer had only considered STING expression in tumor infiltrating inflammatory cells. The study of Menz et al. therefore entirely focused on STING in tumor cells. At least one STING positive case was seen in 130 of 139 tumor types and 96 tumor categories included at least one case with strong positivity. Menz et al described the highest rates of tumor cell STING positivity in squamous cell carcinomas of different sites (up to 96%), malignant mesothelioma (88.5%–95.7%), adenocarcinomas from the pancreas (94.9%), the lung (90.3%), the cervix uteri (90.0%), the colorectum (75.2%), and the gallbladder (68.8%), as well as in serous high-grade ovarian cancer (86.0%). The distribution of positive staining results is shown in an “organ-systematic” and in a “ranking order” figure below (images based on data from Menz et al.). Results on possible associations with histopathological and clinical parameters of tumor aggressiveness are also summarized below (table based on data from Menz et al.).
Authors conclusions on diagnostic utility with respect to the distinction of benign versus malignant (Menz et al.):
- not applicable.
Authors conclusions on diagnostic utility with respect to the distinction of different tumor entities (Menz et al.):
- not applicable.
Authors conclusions on prognostic/predictive role of STING expression (Menz et al.):
- High STING expression was linked to adverse phenotypes in breast cancer, clear cell renal cell carcinoma, colorectal adenocarcinoma, hepatocellular carcinoma, and papillary carcinoma of the thyroid (p<0.05).
- Low STING expression was associated with adverse phenotype (high-grade) in non-invasive urothelial carcinoma (p=0.0002).
Data from the publication: Menz et al. “Stimulator of Interferon Genes Protein (STING) Expression in Cancer Cells: A Tissue Microarray Study Evaluating More than 18,000 Tumors from 139 Different Tumor Entities” Published in Cancers (Basel) 2024, Jun 30 PMID: 39001487
Summarized in own graphics:
Figure1. STING staining in tumors (“organ-specific” ; according to Menz et al.”) with antibody MSVA-515M
Figure1. STING staining in tumors (“ranking order” ; according to Menz et al.”) by positivity with antibody MSVA-515M
Protocol Recommendations
IHC users have different preferences on how the stains should look like. Some prefer high staining intensity of the target stain and even accept some background. Others favor absolute specificity and lighter target stains. Factors that invariably lead to more intense staining include higher concentration of the antibody and visualization tools, longer incubation time, higher temperature during incubation, higher temperature and longer duration of the heat induced epitope retrieval (slide pretreatment). The impact of the pH during slide pretreatment has variable effects and depends on the antibody and the target protein.
All images and data shown here and in our image galleries are obtained by the manual protocol described below. Other protocols resulting in equivalent staining are described as well.
Manual protocol
Freshly cut sections should be used (less than 10 days between cutting and staining). Heat-induced antigen retrieval for 5 minutes in an autoclave at 121°C in pH 7,8 Target Retrieval Solution buffer. Apply MSVA-515M at a dilution of 1:150 at 37°C for 60 minutes. Visualization of bound antibody by the EnVision Kit (Dako, Agilent) according to the manufacturer’s directions.
Potential Research Applications
- The diagnostic utility of STING expression analysis is unclear.
- The prognostic and predictive role of different levels of STING expression in cancer should be investigated.
- A comparison of the expression of STING across different tumor entities would be of interest.
- The function of STING is still not completely clear.
- The utility of STING as a therapeutic target should be evaluated.
Evidence for Antibody Specificity in IHC
There are two ways how the specificity of antibodies can be documented for immunohistochemistry on formalin fixed tissues. These are: 1. Comparison with a second independent method for target expression measurement across a large number of different tissue types (orthogonal strategy), and 2. Comparison with one or several independent antibodies for the same target and showing that all positive staining results are also seen with other antibodies for the same target (independent antibody strategy).
Orthogonal validation: Given the ubiquitous presence of STING RNA across all different normal tissues and the restriction of STING expression to specific cell types, orthogonal validation is not applicable for STING.
Comparison of antibodies: Specificity of MSVA-515M binding for STING is documented by the confirmation of all staining results obtained by MSVA-515M in a parallel analysis using an independent second commercial STING antibody (termed “validation antibody”).