195,00 € – 695,00 €
Product details
Synonyms = BCIE, BIE, EHK, Keratin Type I Cytoskeletal 10, KRT10
Antibody type = Recombinant Mouse monoclonal / IgG1
Clone = MSVA-610M
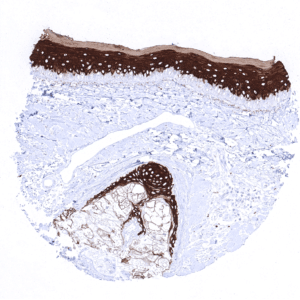
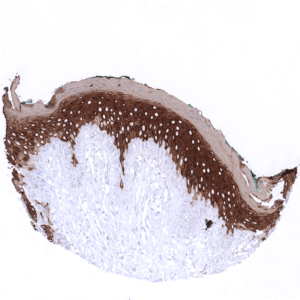
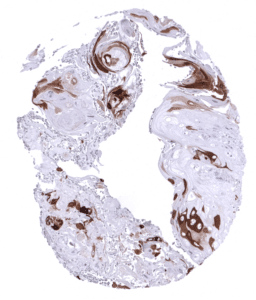
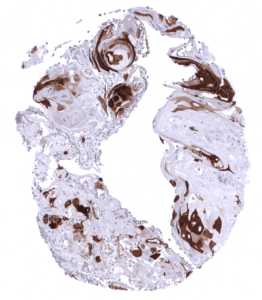
Positive control = Skin: All squamous epithelial cells (except the basal cell layer) should show strong cytoplasmic staining, while the basal cell layer remains KRT10 negative or shows only minimal staining.
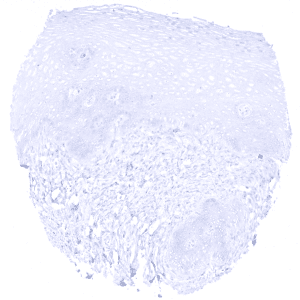
Negative control = Colon: All epithelial, inflammatory, stromal and muscle cells must not show any KRT10 staining.
Cellular localization = Cytoplasmic
Reactivity = Human
Application = Immunohistochemistry
Dilution = 1:100 – 1:200
Intended Use = Research Use Only
Relevance of Antibody
Cytokeratin 10 is expressed in keratinizing squamous epithelial cells
Biology Behind
Cytokeratin 10 (CK10), also termed keratin 10 (KRT10) is a type I acidic high molecular weight keratin protein encoded by the KRT10 gene located 17q21. It dimerizes with the type I keratin 1 and forms intermediate filaments that primarily shape the cytoskeleton of specific epithelial cells. In these cells, KRT10 is part of the cytoskeletal scaffold which contributes to the cell architecture and provides the cells with the ability to withstand mechanical stress. Mutations in CK10 can cause epidermolytic hyperkeratosis or bullous congenital ichthyosiform erythroderma of Brocq.
Staining Pattern in Normal Tissues
Cytokeratin 10 staining pattern in Normal Tissues with antibody MSVA-610M (images are shown in our “Normal Tissue Gallery”)
| Brain | Cerebrum | Negative. |
| Cerebellum | Negative. | |
| Endocrine Tissues | Thyroid | Negative. |
| Parathyroid | Negative. | |
| Adrenal gland | Negative. | |
| Pituitary gland | Negative. | |
| Respiratory system | Respiratory epithelium | Negative. |
| Lung | Negative. | |
| Gastrointestinal Tract | Salivary glands | Negative. |
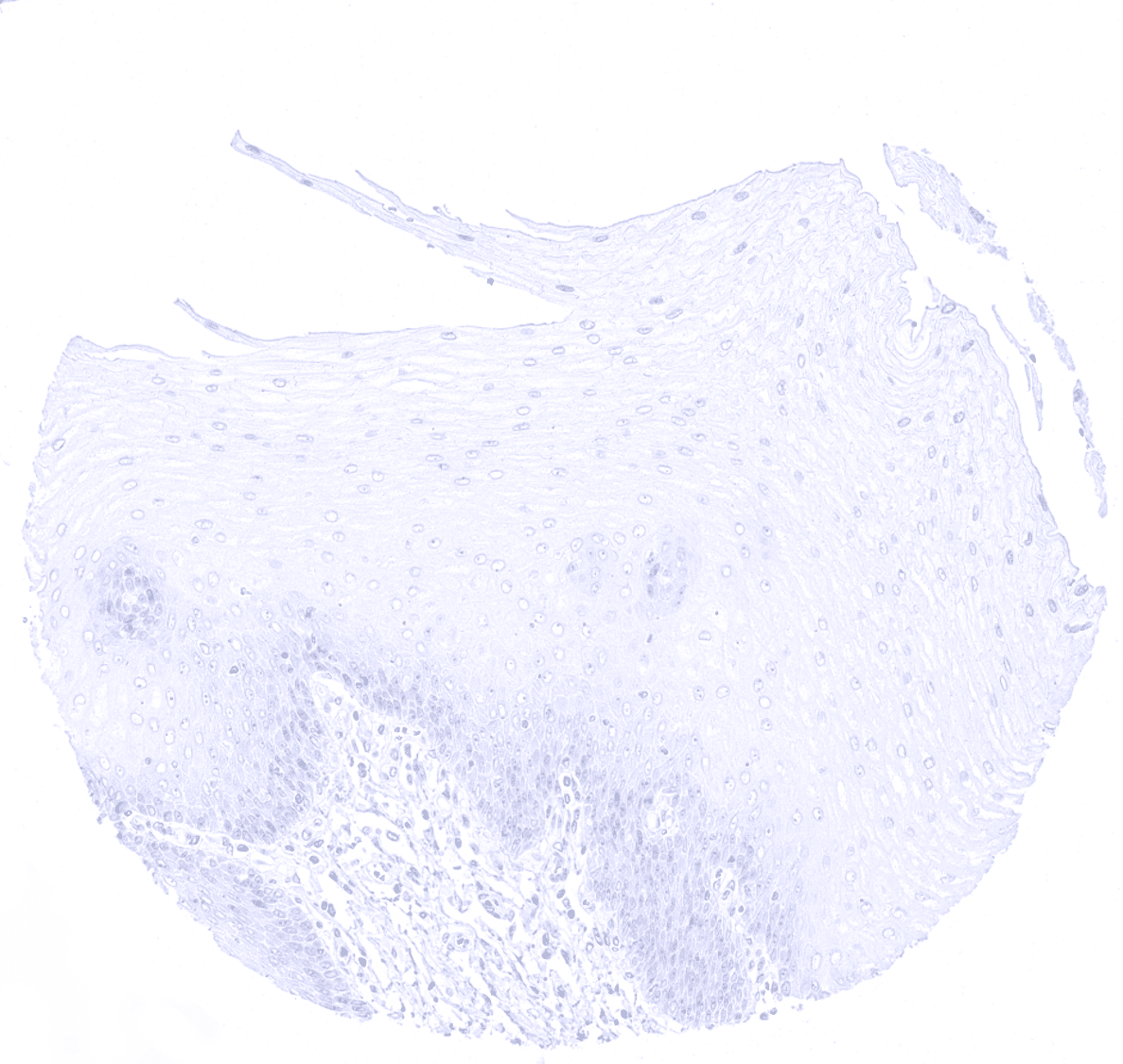
| Esophagus | Usually negative. | |
| Stomach | Negative. | |
| Duodenum | Negative. | |
| Small intestine | Negative. | |
| Appendix | Negative. | |
| Colon | Negative. | |
| Rectum | Negative. | |
| Liver | Negative. | |
| Gallbladder | Negative. | |
| Pancreas | Negative. | |
| Genitourinary | Kidney | Negative. |
| Urothelium | Negative. | |
| Male genital | Prostate | Negative. |
| Seminal vesicles | Negative. | |
| Testis | Negative. | |
| Epididymis | Negative. | |
| Female genital | Breast | Negative. |
| Uterus, myometrium | Negative. | |
| Uterus, ectocervix | Usually negative. | |
| Uterus endocervix | Negative. | |
| Uterus, endometrium | Negative. | |
| Fallopian Tube | Negative. | |
| Ovary | Negative. | |
| Placenta early | Negative. | |
| Placenta mature | Negative. | |
| Amnion | Negative. | |
| Chorion | Negative. | |
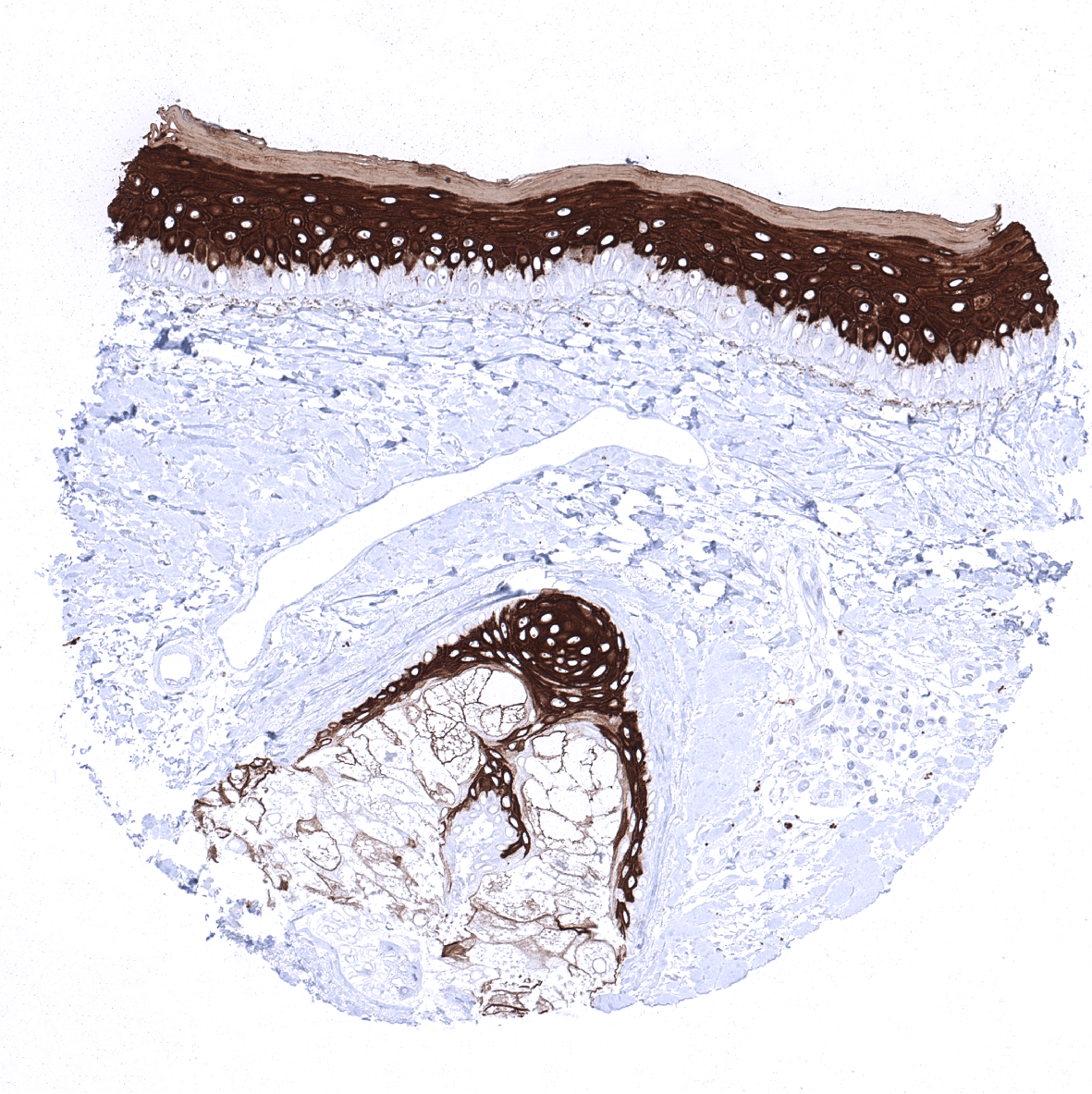
| Skin | Epidermis | Strong CK10 staining of all suprabasal cell layers of the keratinizing squamous epithelium. The basal cell layer of the epidermis is CK10 negative. |
| Sebaceous glands | Moderate to strong CK10 staining sebaceous glands and suprabasal cells of hair follicles. | |
| Muscle/connective tissue | Heart muscle | Negative. |
| Skeletal muscle | Negative. | |
| Smooth muscle | Negative. | |
| Vessel walls | Negative. | |
| Fat | Negative. | |
| Stroma | Negative. | |
| Endothelium | Negative. | |
| Bone marrow/lymphoid tissue | Bone marrow | Negative. |
| Lymph node | Negative. | |
| Spleen | Negative. | |
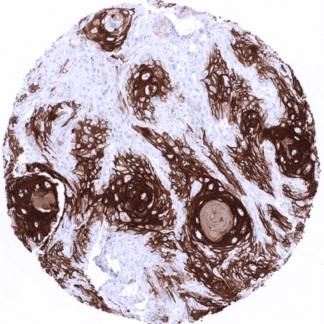
| Thymus | Moderate to strong CK10 staining of keratinizing squamous epithelial cells the central areas of corpuscles of Hassall’s. | |
| Tonsil | Moderate to strong CK10 staining of a fraction of superficial keratinizing squamous epithelial cells in the of tonsil crypts. | |
| Remarks |
CK10 is always strongly expressed in all suprabasal cell layers of the keratinizing squamous epithelium of the skin as well as in associated sebaceous glands and hair follicles. The basal cell layer of the epidermis is strictly CK10 negative. Strong CK10 expression is also regularly observed in a superficial keratinizing fraction of cells in the squamous epithelium of tonsil crypts and the central areas of corpuscles of Hassall’s. CK10 is usually absent in non-keratinizing squamous epithelium such as of the ectocervix, tonsil surface, esophagus, oral cavity and the lip. However, a variable fraction of suprabasal cells or groups of cells in these non-keratinizing squamous epithelia may occasionally show a weak, moderate, or even strong KRT10 staining. KRT10 immunostaining is absent in urothelium and transitional epithelium of the anal mucosa. CK10 immunostaining was absent in gastrointestinal epithelium, gallbladder, liver, pancreas, lung, fallopian tube, endocervical glands of the uterus, ovary, placenta, lymphatic, hematopoetic and mesenchymal tissues, the pituitary gland, and the brain.
These findings are largely comparable to the RNA and protein data summarized in the Human Protein Atlas (Tissue expression Cytokeratin 10)
Suggested positive tissue control: Skin: All squamous epithelial cells (except the basal cell layer) should show strong cytoplasmic staining, while the basal cell layer remains KRT10 negative or shows only minimal staining.
Suggested negative tissue control: Colon: All epithelial, inflammatory, stromal and muscle cells must not show any KRT10 staining.
Staining Pattern in Relevant Tumor Types
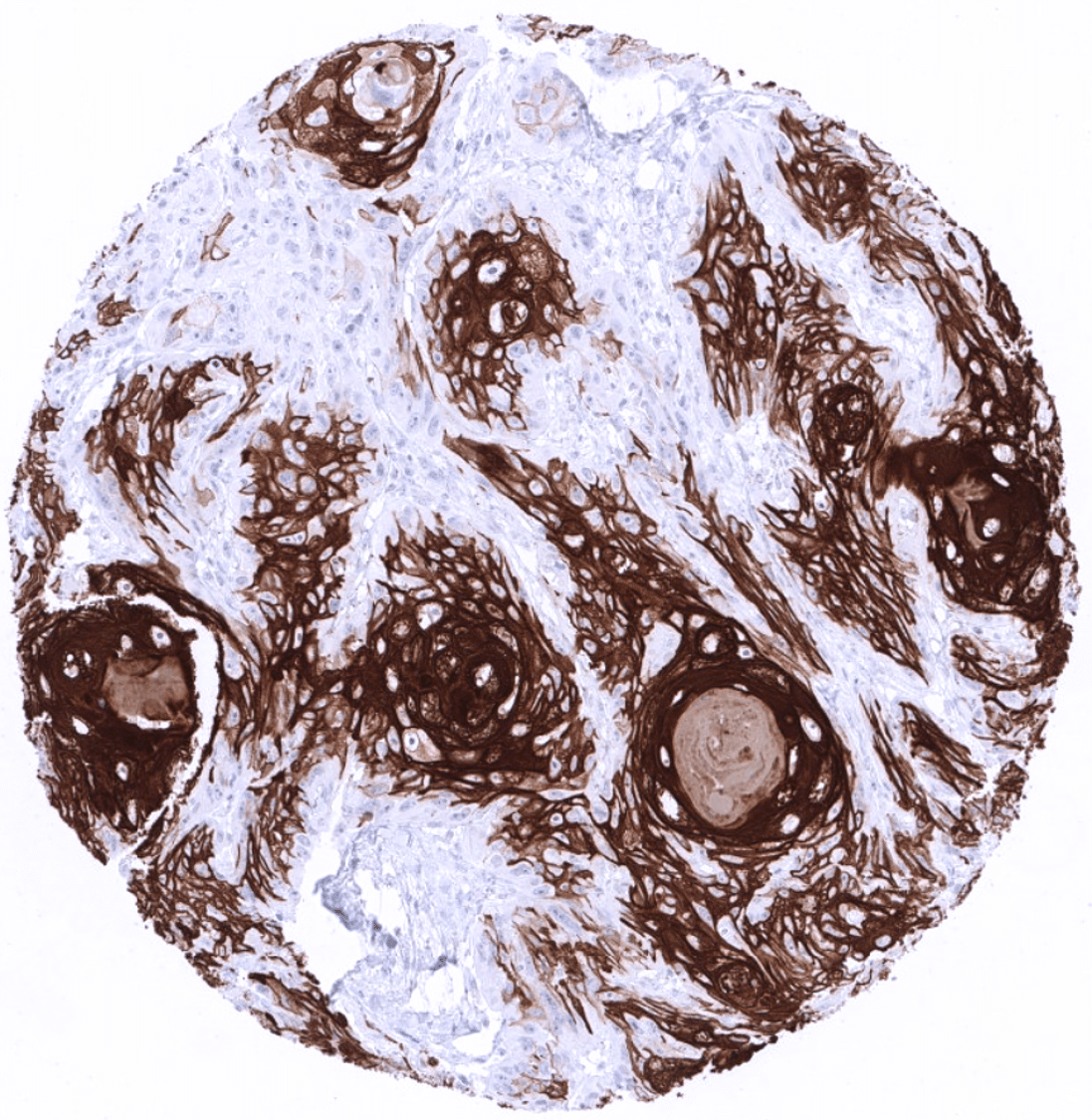
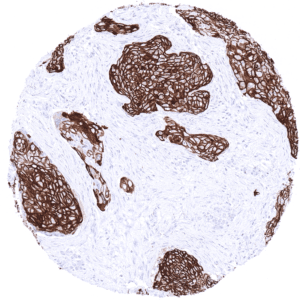
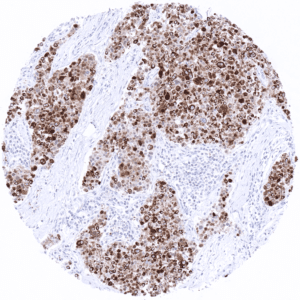
KRT10 immunostaining almost exclusively occurs in squamous cell carcinomas of various sites of origin. The rate of KRT10 positive cases may vary depending on the histologic tumor grade. Some of the KRT10 positive squamous cell carcinomas show – similar to the situation in normal skin – absence of expression in the basal cell layers.
The TCGA findings on Cytokeratin 10 RNA expression in different tumor categories have been summarized in the Human Protein Atlas.
Compatibility of Antibodies
Cytokeratin 10 (CK10) (MSVA-610M) publication summary
Relevant publication: Uhlig et al.: “Cytokeratin 10 (CK10) expression in cancer: A tissue microarray study on 11,021 tumors”. Published in Ann Diagn Pathol. 2022 Aug 20;60:152029. Epub ahead of print. PMID: 36029589.
A total of 9’462 tumors were successfully analyzed from 131 different tumor categories by using the following protocol: Heat-induced antigen retrieval for 5 minutes in an autoclave at 121°C in pH7,8 Target Retrieval Solution buffer. MSVA-610M at a dilution of 1:150 at 37°C for 60 minutes. Visualization of bound antibody by the EnVision Kit (Dako, Agilent). This protocol was also used for all stainings depicted in our tumor and normal tissue galleries.
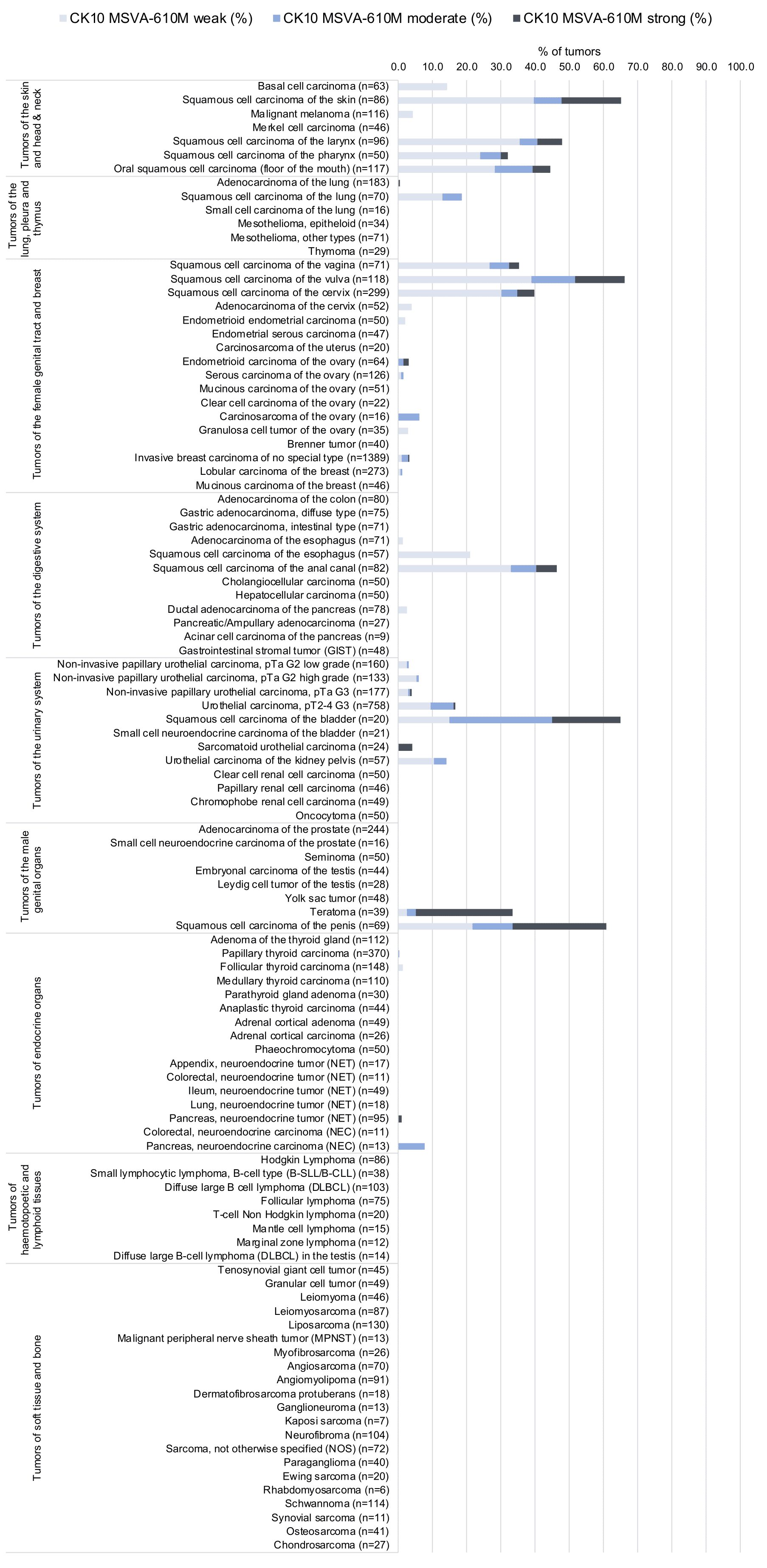
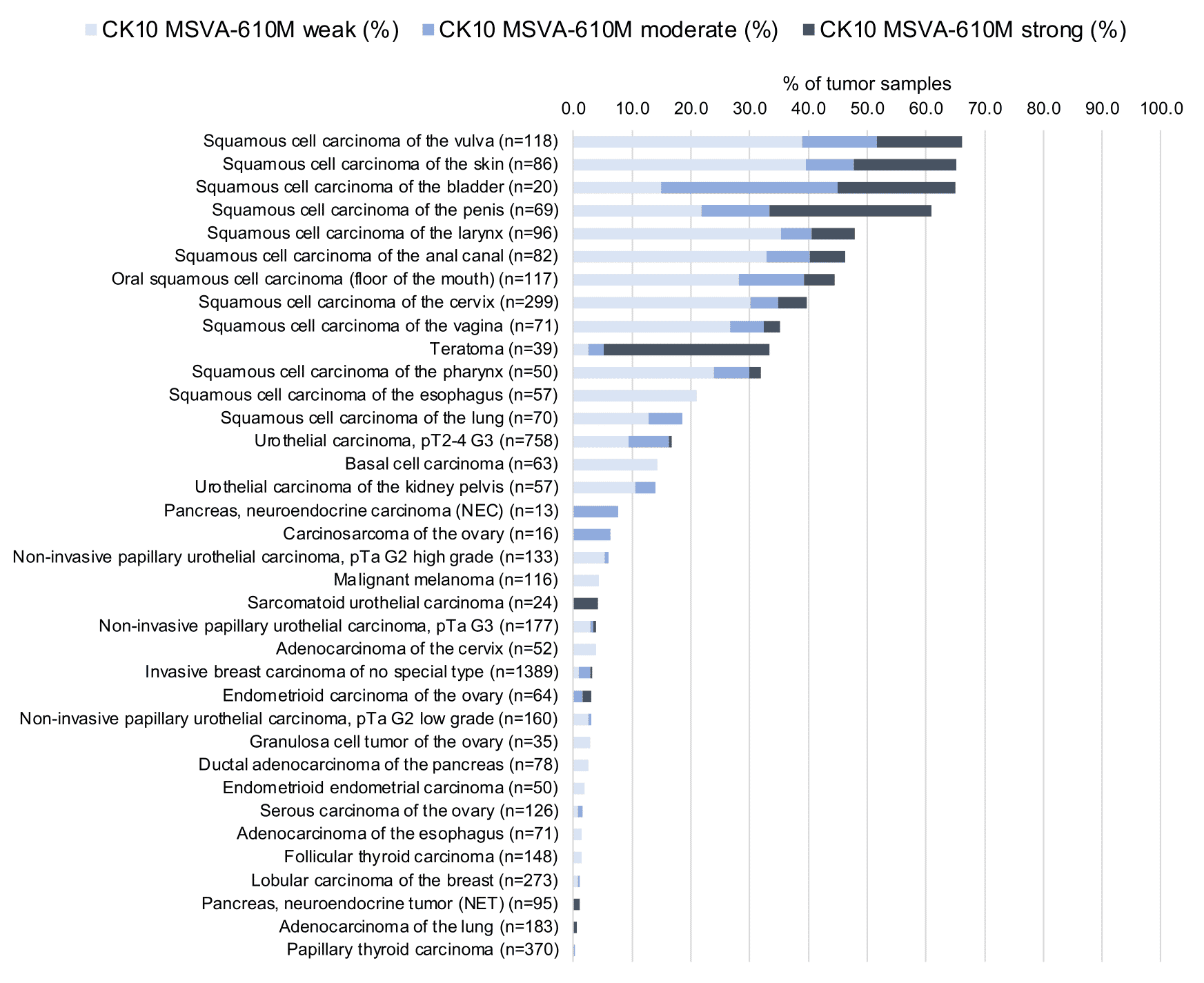
In this study, at least one CK10 positive case was seen in 41 (31,3%) of 131 tumor categories and 18 (13,7%) tumor categories included at least one case with strong positivity. Uhlig et al described the highest CK10 positivity rates in squamous cell carcinomas from various sites (positive in 18.6–66.1%) and in Warthin tumors of salivary glands (47.8%), followed by various tumor entities known to potentially exhibit areas with squamous cell differentiation such as teratomas (33.3%), basal cell carcinomas of the skin (14.3%), adenosquamous carcinomas of the cervix (11.1%), and several urothelial neoplasms (3.1–16.8%). The CK staining was often intense but limited to areas of mature squamous epithelium undergoing keratinization. This limitation of even strong CK10 staining to rather small subsets of tumor cells resulted in most positive tumors being classified as only weakly or moderately positive according to the predefined criteria of Uhlig et al. In a small fraction of cases, CK10 positivity was also described in tumors without obvious squamous cell morphology. The distribution of positive staining results is shown in an “organ-systematic” and in a “ranking order” figure below (images based on data from Uhlig et al.). Results on possible associations with histopathological and clinical parameters of tumor aggressiveness are also summarized below (table based on data from Uhlig et al.).
Authors conclusions on diagnostic utility of CK10 immunohistochemistry with respect to the distinction of neoplastic from non-neoplastic tissues (Uhlig et al.):
- Reports from other authors have suggested that aberrant expression of CK10 in non-keratinizing squamous epithelium may be a feature of malignant transformation.
Authors conclusions on diagnostic utility of CK10 immunohistochemistry with respect to the distinction of different tumor entities (Uhlig et al.):
- CK10 as a suitable marker for mature, keratinizing squamous differentiation in epithelial tissues.
- Detectable CK10 immunostaining may be indicative for squamous cell differentiation in urothelial neoplasms.
Authors conclusions on prognostic/predictive role of CK10 expression (Uhlig et al.):
- Absence of CK10 staining was linked to high stage (p=0,0032) and poor grade (p<0,0001) in a collection of squamous cell carcinomas from different sites of origin.
- CK10 IHC may be instrumental for a more objective evaluation of the clinical significance of focal squamous differentiation in cancer.
Figure 1. Cytokeratin 10 staining in cancer (“organ-systematic”; according to Uhlig et al.)
Figure 2. Cytokeratin 10 staining in cancer (“ranking list”; according to Uhlig et al.)
Protocol Recommendations
IHC users have different preferences on how the stains should look like. Some prefer high staining intensity of the target stain and even accept some background. Others favor absolute specificity and lighter target stains. Factors that invariably lead to more intense staining include higher concentration of the antibody and visualization tools, longer incubation time, higher temperature during incubation, higher temperature and longer duration of the heat induced epitope retrieval (slide pretreatment). The impact of the pH during slide pretreatment has variable effects and depends on the antibody and the target protein. Accordingly, multiple different protocols can generate identical staining results.
All images and data shown here and in our image galleries were obtained by the manual protocol described below. Other protocols resulting in equivalent staining are described as well.
Manual protocol
Freshly cut sections should be used (less than 10 days between cutting and staining). Heat-induced antigen retrieval for 5 minutes in an autoclave at 121°C in pH7,8 Target Retrieval Solution buffer. Apply MSVA-610M at a dilution of 1:150 at 37°C for 60 minutes. Visualization of bound antibody by the EnVision Kit (Dako, Agilent) according to the manufacturer’s directions.
Impact of pH
MSVA-610M results in strongest staining if pH9,0 is used for slide pretreatment. pH7,8 is acceptable but lower pH results in a significant reduction of sensitivity.
Potential Research Applications
- The diagnostic utility of reduced CK10 expression in the skin is not known.
- The diagnostic utility of CK10 neo-expression in non-keratinizing squamous epithelium of ectocervix, esophagus, oral cavity and lip is not known.
- The prognostic role of CK10 expression in squamous cell carcinoma is unknown.
Evidence for Antibody Specificity in IHC
Utility of MSVA-610M is documented by a staining pattern that exactly matches the data summarized in the protein atlas. A strong positive KRT10 staining almost exclusively seen in the skin. All other tissues showing weak or occasional KRT10 staining are also squamous epithelia. In addition, all tissues known to not express KRT10 including those notorious for non-specific IHC background such as kidney and colonic mucosa are completely KRT10 negative.