395,00 € – 1.495,00 €
Product details
Synonyms = Carboxypeptidase A1 (pancreatic); CPA1; Pancreatic Carboxypeptidase A1; Procarboxypeptidase A1 pancreatic
Antibody type = Mouse monoclonal / IgG1
Clone = MSVA-601M
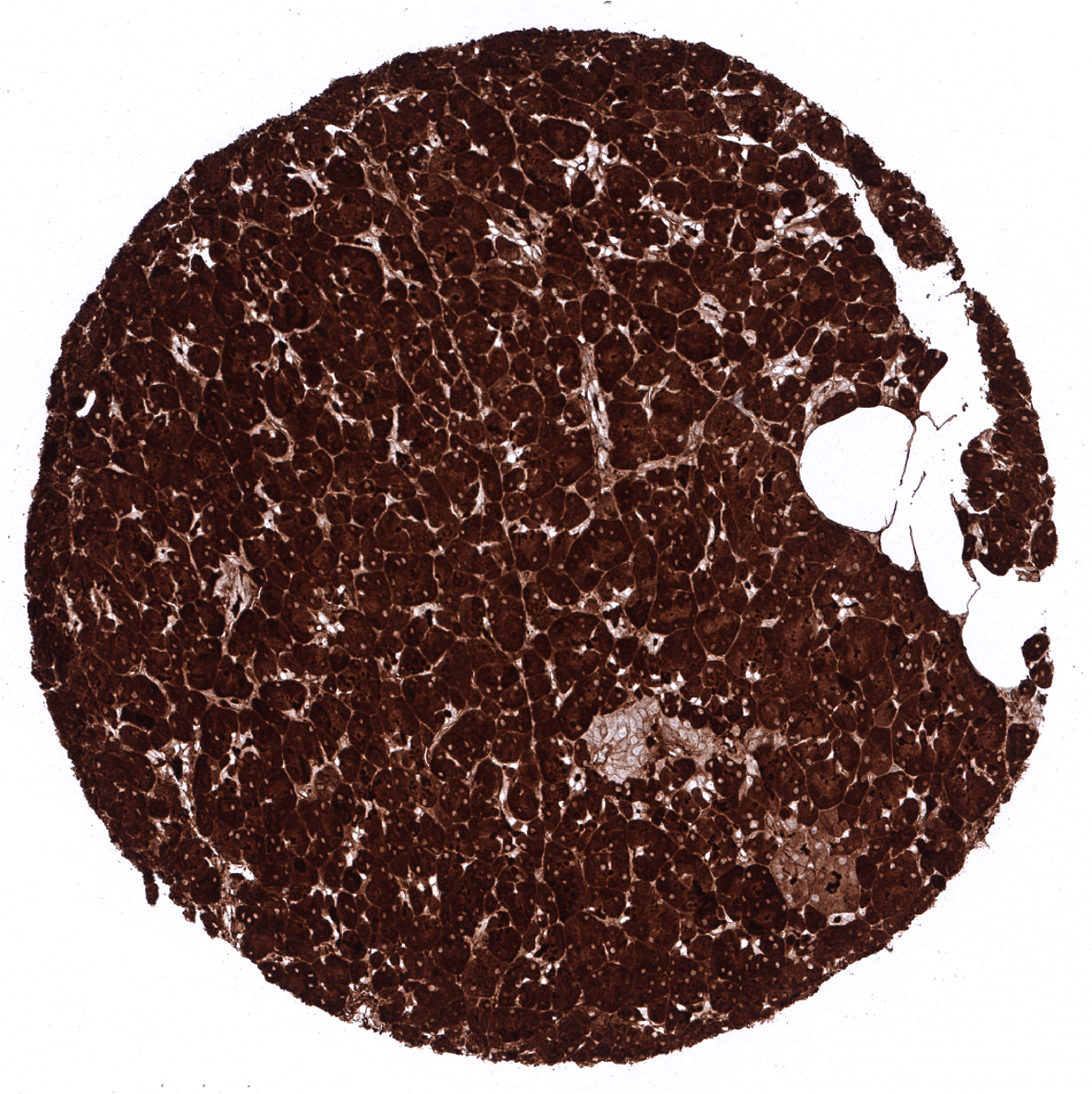
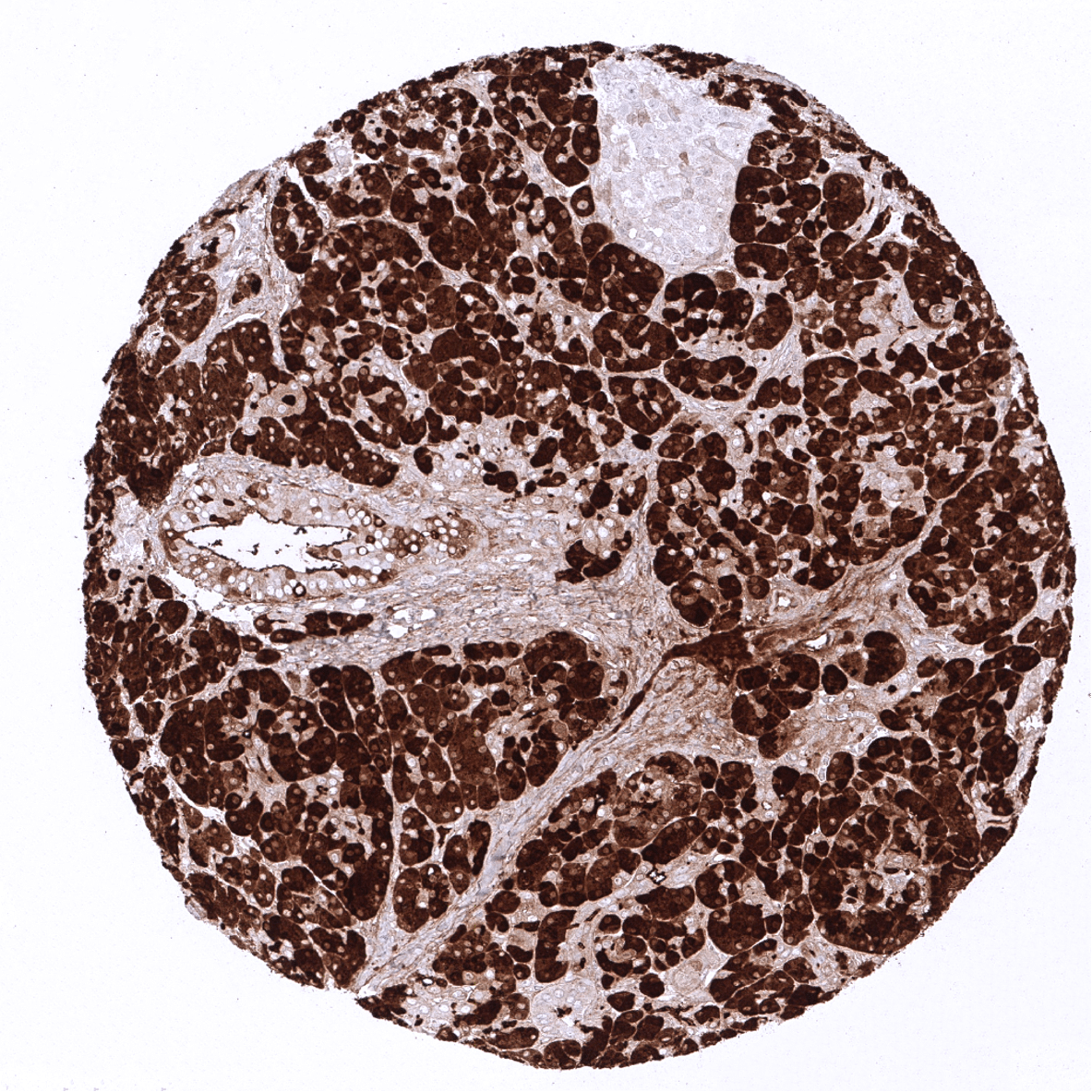
Positive control = Pancreas: A strong CPA1 staining of acinar cells should be seen. Adjacent structures can also be stained due to contamination artifacts.
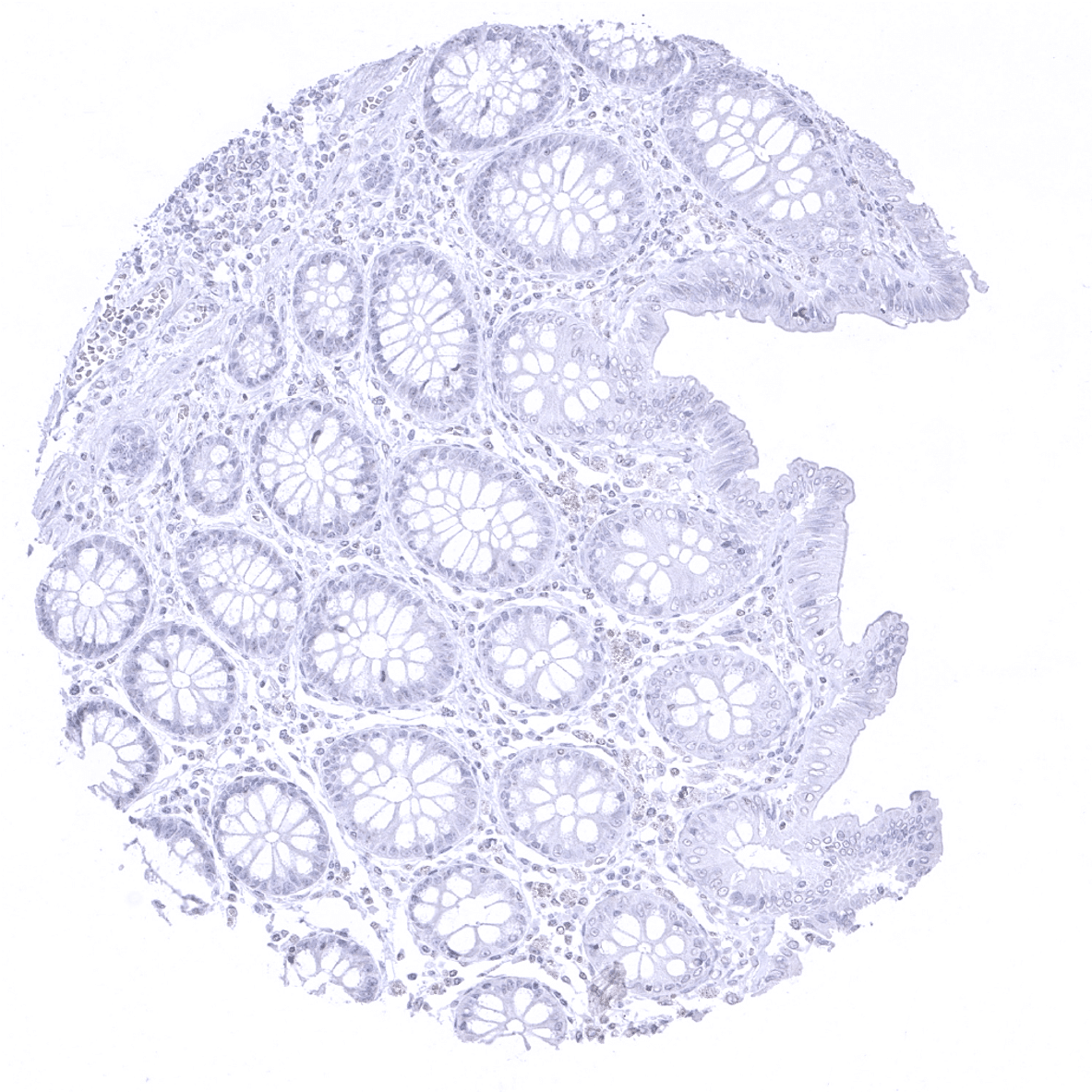
Negative control = Colon: CPA1 immunostaining should not be seen.
Cellular localization = Cytoplasmic and secreted.
Reactivity = Human
Application = Immunohistochemistry
Dilution = 1:100 – 1:200
Intended Use = Research Use Only
Relevance of Antibody
CPA1 is expressed on normal and neoplastic pancreatic acinar cells.
Recently presented poster at the USCAP Annual Meeting in Los Angeles March 19-24th 2022 by Ria Uhlig.
“Suitability of combined immunohistochemistry analysis of CPA1 and CELA3B for diagnosing acinar cell carcinoma of the pancreas.”[1]
[1] Poster Session, March 22, 2022 USCAP Annual Meeting 2022; Los Angeles Convention Center in West Exhibit Hall A, Poster Board #:164
Presenting author: Ria Uhlig (Department of Pathology, University Medical Center Hamburg – Eppendorf, Hamburg, Germany)
Biology Behind
Carboxypeptidase A1 (CPA1) is a zinc metalloprotease coded by the CPA1 gene located at 7q32.2. It is a 34,6kDa protein which is solely produced in the pancreas. It is involved in zymogen inhibition and was shown to preferentially cleave C-terminal branched-chain and aromatic amino acids from dietary proteins. Mutations of CPA1 gene have been linked to chronic pancreatitis. Elevated CPA1 serum protein levels have been described in patients with pancreatic cancer. [1]
[1] Uhlig R et al. “Carboxypeptidase A1 (CPA1) Immunohistochemistry Is Highly Sensitive and Specific for Acinar Cell Carcinoma (ACC) of the Pancreas.” Am J Surg Pathol. 2022 Jan 1;46(1):97-104. PMID: 34889867.
Staining Pattern in Normal Tissues
CPA1 staining pattern in Normal Tissues with antibody MSVA-601M (images are shown in our “Normal Tissue Gallery”)
| Brain | Cerebrum | Negative. |
| Cerebellum | Negative. | |
| Endocrine Tissues | Thyroid | Negative. |
| Parathyroid | Negative. | |
| Adrenal gland | Negative. | |
| Pituitary gland | Negative. | |
| Respiratory system | Respiratory epithelium | Negative. |
| Lung | Negative. | |
| Gastrointestinal Tract | Salivary glands | Negative. |
| Esophagus | Negative. | |
| Stomach | Negative. | |
| Colon | Negative. | |
| Duodenum | Negative. | |
| Rectum | Negative. | |
| Small intestine | Negative. | |
| Liver | Negative. | |
| Gallbladder | Negative. | |
| Pancreas | Strong cytoplasmic CPA1 staining in all acinar cells. CPA1 does not seem to be expressed in islet cells, intercalated ducts and excretory ducts – but all these cell types can show a variable intensity staining due to contamination artifacts caused by high levels of CPA1 in adjacent acinar cells. | |
| Genitourinary | Kidney | Negative. |
| Urothelium | Negative. | |
| Male genital | Prostate | Negative. |
| Seminal vesicles | Negative. | |
| Testis | Negative. | |
| Epididymis | Negative. | |
| Female genital | Breast | Negative. |
| Uterus, ectocervix | Negative. | |
| Uterus endocervix | Negative. | |
| Uterus, endometrium | Negative. | |
| Fallopian Tube | Negative. | |
| Ovary | Negative. | |
| Placenta early | Negative. | |
| Placenta mature | Negative. | |
| Amnion | Negative. | |
| Chorion | Negative. | |
| Skin | Epidermis | Negative. |
| Sebaceous glands | Negative. | |
| Muscle/connective tissue | Heart muscle | Negative. |
| Skeletal muscle | Negative. | |
| Smooth muscle | Negative. | |
| Fat | Negative. | |
| Bone marrow/lymphoid | Bone marrow | Negative. |
| Lymph node | Negative. | |
| Spleen | Negative. | |
| Thymus | Negative. | |
| Tonsil | Negative. | |
| Remarks | The CPA1 expression level in normal acinar cells is very high. Therefore, acinar-cell adjacent structures can show “pseudo-positivity” due to an artificial spread of CPA1 protein from neighboring acinar cells. |
These findings are largely comparable to the RNA and protein data described in the Human Protein Atlas (Tissue expression CPA1).
Suggested positive tissue control: Pancreas: A strong staining of acinar cells should be seen. Adjacent structures can also be stained due to contamination artifacts.
Suggested negative tissue control: Colon: CPA1 immunostaining must be absent in all cell types.
Staining Pattern in Relevant Tumor Types
CPA1 immunostaining is predominantly seen in acinus cell carcinomas of the pancreas.
The TCGA findings on CPA1 RNA expression in different tumor categories have been summarized in the Human Protein Atlas.
Compatibility of Antibodies
CPA1 (MSVA-601M) publication summary:
Relevant publication: Uhlig et al. “Carboxypeptidase A1 (CPA1) Immunohistochemistry is Highly Sensitive and Specific for Acinar Cell Carcinoma (ACC) of the Pancreas”. Published in the American Journal of Surgical Pathology. 2022 Jan 1;46(1):97-104.
A total of 12,274 tumors were analyzed from 131 different tumor categories by using the following protocol: Heat-induced antigen retrieval for 5 minutes in an autoclave at 121°C in pH7,8 Target Retrieval Solution buffer. MSVA-601M at a dilution of 1:150 at 37°C for 60 minutes. Visualization of bound antibody by the EnVision Kit (Dako, Agilent). This protocol was also used for all stainings depicted in our tumor and normal tissue galleries.
A positive CPA1 immunostaining was seen in all 12 analyzed pancreatic acinar cell carcinomas but in none of 12263 tumors from 130 other tumor categories. The distribution of positive staining results is shown in an “organ-systematic” figure below (images based on data from Uhlig et al.).
Authors conclusions on diagnostic utility with respect to the distinction of benign versus malignant (Uhlig et al.):
- not applicable.
Authors conclusions on diagnostic utility with respect to the distinction of different tumor entities (Uhlig et al.):
- CPA1 is a highly sensitive and specific marker for acinar cell carcinoma of the pancreas.
Authors conclusions on prognostic/predictive role of CPA1 expression.
- not applicable.
Data from the publication: “Carboxypeptidase A1 (CPA1) Immunohistochemistry Is Highly Sensitive and Specific for Acinar Cell Carcinoma (ACC) of the Pancreas.” Published by Uhlig R et al. in the American Journal of Surgical Pathology. 2022 Jan 1;46(1):97-104.
Summarized in own graphic:
1. CPA1 staining in tumors “organ-specific” with antibody MSVA-601M
Protocol Recommendations
IHC users have different preferences on how the stains should look like. Some prefer high staining intensity of the target stain and even accept some background. Others favor absolute specificity and lighter target stains. Factors that invariably lead to more intense staining include higher concentration of the antibody and visualization tools, longer incubation time, higher temperature during incubation, higher temperature and longer duration of the heat induced epitope retrieval (slide pretreatment). The impact of the pH during slide pretreatment has variable effects and depends on the antibody and the target protein.
All images and data shown here are obtained by the manual protocol described below. Protocols resulting in equivalent staining by using an automated immunostainer are described as well. Visualization of bound antibody by the EnVision Kit (Dako, Agilent) according to the manufacturer’s directions.
Manual protocol
Freshly cut sections should be used (less than 10 days between cutting and staining). Heat-induced antigen retrieval for 5 minutes in an autoclave at 121°C in pH 7,8 Target Retrieval Solution buffer. Apply MSVA-601M at a dilution of 1:150 at 37°C for 60 minutes. Visualization of bound antibody by the EnVision Kit (Dako, Agilent) according to the manufacturer’s directions.
Agilent / Dako – Autostainer Link 48
Pretreatment in PT-Link for 30 minutes at 95°C (pH high); FLEX peroxidase blocking for 5 minutes (room temperature), MSVA-601M 1:150 for 20 minutes (room temperature), FLEX+ mouse/rabbit (LINKER) for 15 minutes (room temperature), horseradish peroxidase (HRP) for 20 minutes (room temperature), FLEX DAB+Sub-Chromo for 10 minutes (room temperature), FLEX hematoxylin for 5 minutes (room temperature).
These images reflect stainings by the protocol described above. It is of note that a comparable staining result can also be obtained by different protocols. In general, a longer pretreatment, a longer incubation time of the primary antibody, a higher antibody concentration, and a longer incubation time of FLEX+LINKER result in stronger staining, potentially at the cost of more background staining. Modifications of the protocol with a strengthening effect on staining intensity in combination with changes of other parameters that result in lower staining intensity can result in a comparable result as shown above.
Leica – BOND RX
Dewax at 72°C for 30 seconds; Pretreatment in Bond Epitope Retrieval Solution (ER2 – EDTA pH9) for 40 minutes at 100°C; Peroxidase blocking for 5 minutes (room temperature), MSVA-601M 1:300 for 20 minutes (room temperature), Post primary (rabbit anti mouse) for 8 minutes (room temperature), Polymer (goat anti rabbit) for 8 minutes (room temperature), mixed DAB refine for 10 minutes (room temperature), hematoxylin for 5 minutes (room temperature).
These images reflect stainings by the protocol described above. It is of note that a comparable staining result can also be obtained by different protocols. In general, a longer pretreatment, a longer incubation time of the primary antibody, a higher antibody concentration, a higher temperature during incubation, and a longer incubation time of Post primary and or the Polymer result in stronger staining, potentially at the cost of more background staining. Modifications of the protocol with a strengthening effect on staining intensity in combination with changes of other parameters that result in lower staining intensity can result in a comparable result as shown above.
Roche – Ventana Discovery ULTRA
Pretreatment for 64 minutes at 100°C (pH 8,4); CM peroxidase blocking for 12 minutes (room temperature), MSVA-601M 1:150 for 20 minutes at 36°C, secondary antibody (anti-mouse HQ) for 12 minutes at 36°C, anti-HQ HRP for 12 minutes at room temperature, DAB at room temperature, hematoxylin II at room temperature for 8 minutes, bluing reagent at room temperature for 4 minutes.
These images depict staining results obtained by the protocol described above. It is of note, that the Ventana machines generally require higher antibody concentrations than other commonly used autostainers because the antibodies are automatically diluted during the procedure. Various other protocols can result in an identical result as shown above. A longer pretreatment, a longer incubation time of the primary antibody, a higher antibody concentration, a higher temperature during incubation, and a longer incubation time of secondary antibody and or the anti-HQ HRP result in stronger staining, potentially at the cost of more background staining.
Potential Research Applications
- CPA1 expression has been shown to represent a specific feature of pancreatic acinar cell carcinoma. Further studies are needed to validate this findings.
- The prognostic relevance of different CPA1 expression levels should be investigated in pancreatic acinar cell carcinoma.
- Can elevated CPA1 serum levels already suggest acinar cell carcinoma
Evidence for Antibody Specificity in IHC
There are two ways, how the specificity of antibodies can be documented for immunohistochemistry on formalin fixed tissues. These are: 1. comparison with a second independent method for target expression measurement across a large number of different tissue types (orthogonal strategy), and 2. Comparison with one or several independent antibodies for the same target and showing that all positive staining results are also seen with other antibodies for the same target (independent antibody strategy).
For the antibody MSVA-601M specificity is documented by the strong concordance of the immunostaining with RNA expression data derived from the Human Protein Atlas (HPA) RNA-seq tissue dataset, the FANTOM5 project, and the Genotype-Tissue Expression (GTEx) project which are all summarized in the Human Protein Atlas (Tissue expression CPA1). Immunostaining by using MSVA-601M was exclusively detected in the pancreas, the only organ with documented RNA expression.
Moreover, no staining was seen in tissues notorious for non-specific IHC background such as kidney, colonic mucosa, and epidermis.