295,00 € – 895,00 €
Product details
Synonyms = Allograft inflammatory factor 1 (AIF1); balloon angioplasty responsive transcription (BART1); IBA1; Interferon responsive transcript 1; Ionized calcium-binding adapter molecule 1; IRT1; Microglia response factor (MRF1)
Antibody type = Mouse monoclonal / IgG
Clone = MSVA-955M
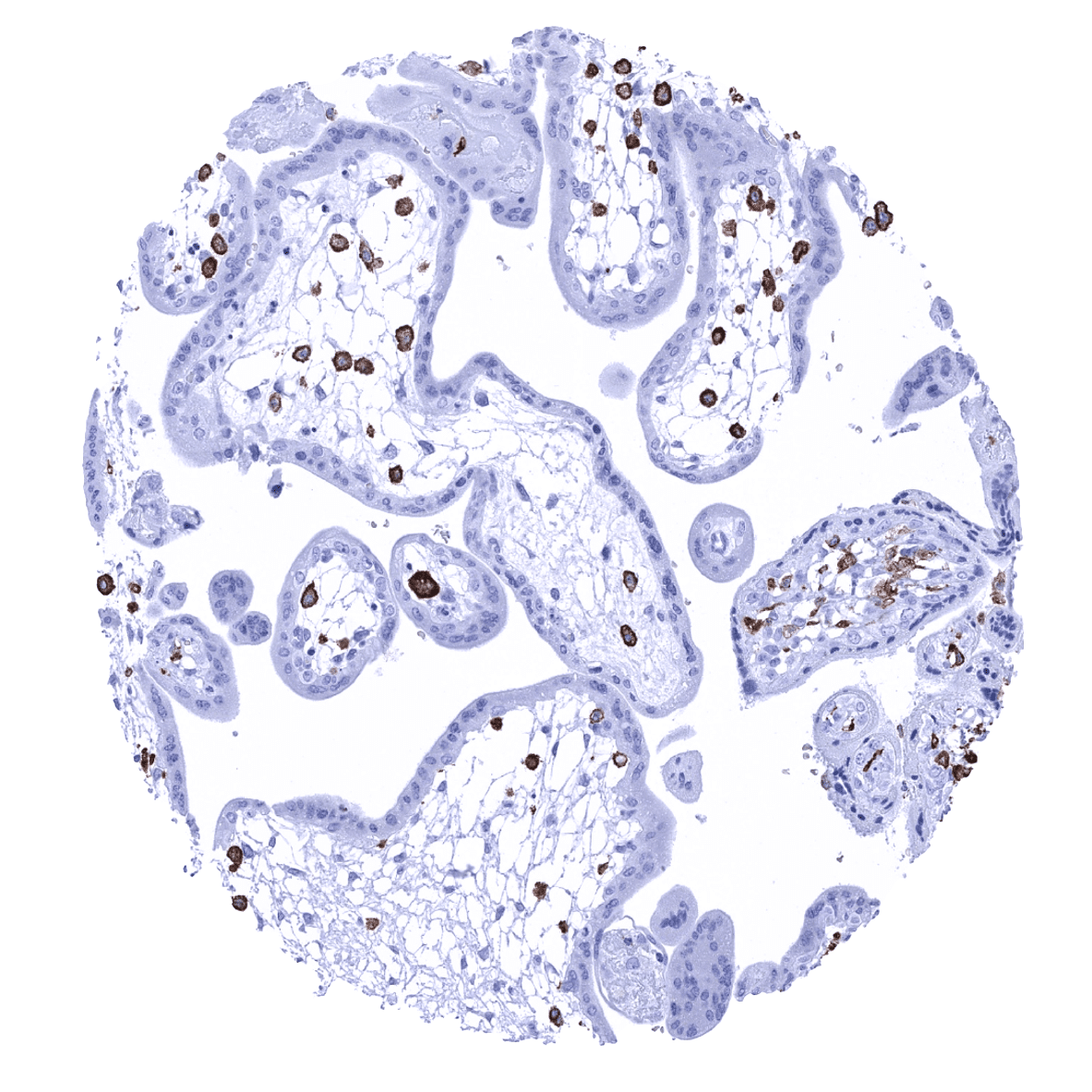
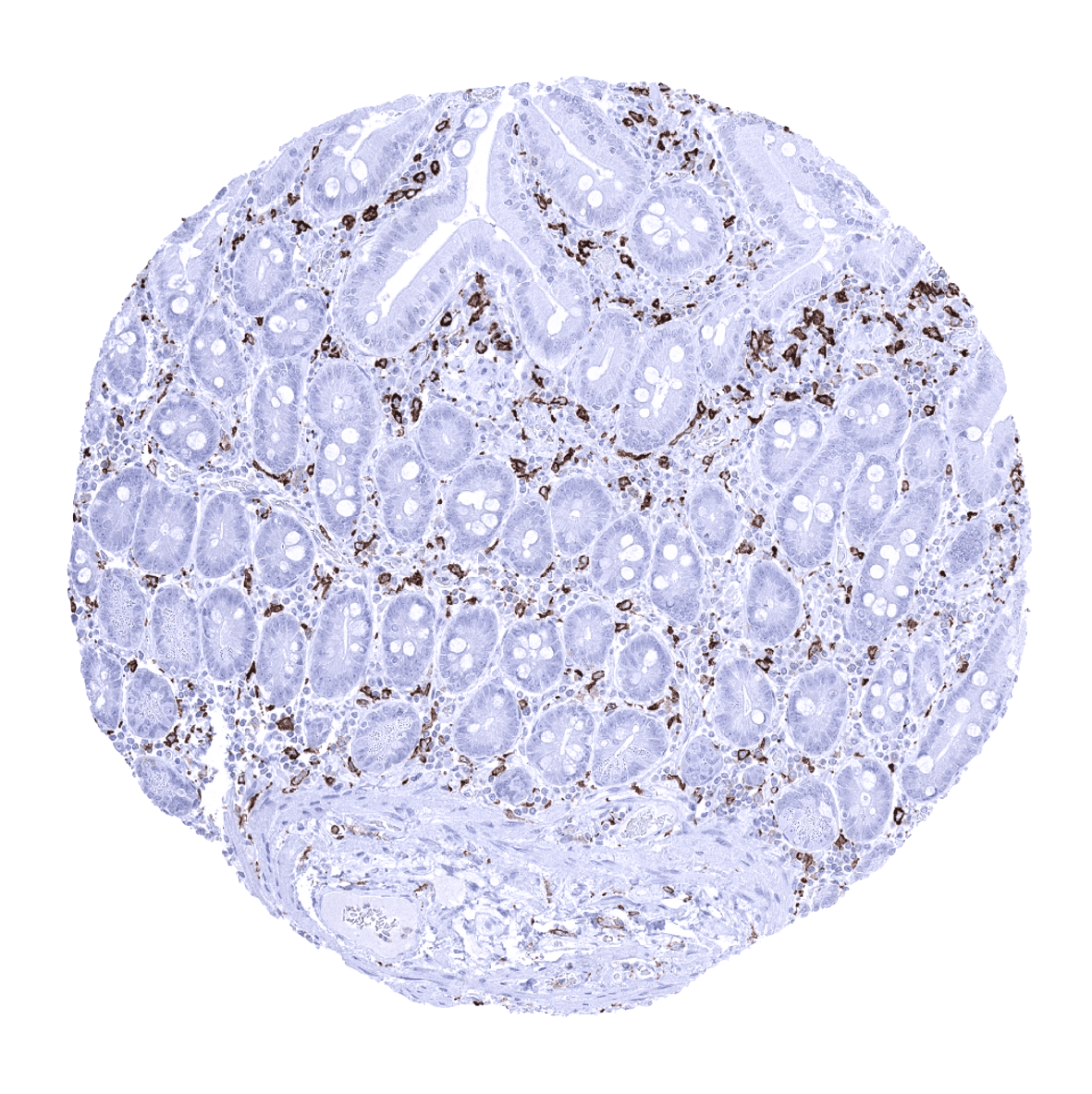
Positive control = Colon: An at least moderate to strong cytoplasmic AIF1 immunostaining should be seen in mucosal macrophages.
Negative control = Colon: AIF1 immunostaining should be absent in all non-inflammatory cells.
Cellular localization = Cytoplasmic and cell surface.
Reactivity = Human
Application = Immunohistochemistry
Dilution = 1:100 – 1:200
Intended Use = Research Use Only
Relevance of Antibody
AIF1 is a putative marker for macrophage activation status.
Biology Behind
Allograft inflammatory factor 1 (AIF1) is coded by the AIF1 gene at 6p21.3 where it is located within the major histocompatibility complex class III region known to harbor clusters of genes involved in inflammatory responses. AIF1 is a phylogenetically conserved gene. Three isoforms exist, the largest of which, AIF1 splice variant 3 (AIF1v3), has a molecular mass of 17 kDa. The function of AIF1 is not well known. The protein is upregulated in activated macrophages and neutrophils in response to the cytokine IFN-γ. AIF1 impacts the expression of several important mediators such as cytokines, chemokines and inducible nitric oxide synthase. It is involved in inflammatory responses, auto-immune diseases, reproductive immunity as well as immune activation and macrophage function. AIF1 may also regulate several important cell adhesion molecules. Increased AIF1 expression levels have been found in cancers and AIF1 has been suggested to have a significant role in cancer progression.
Staining Pattern in Normal Tissues
AIF1 immunostaining is seen at varying levels of intensity in histiocytes/macrophages in various different tissues. The staining is mostly moderate to strong but varies depending on the location of cells and also between different samples from identical tissues. For example, AIF1 immunostaining is considerably weaker in dendritic cells/macrophages of the germinal centre than in macrophages of the interfollicular area in lymph nodes and tonsils. AIF1 expression can be particularly strong in Kupffer cells in the liver, placenta macrophages (variable between samples), lung, and the adrenal gland. In the brain, a variable staining of microglia occurs, ranging from low to high intensity. Granulocytes also stain AIF1 positive. In the kidney, a moderate to strong membranous staining of glomeruli is seen. This reflects the only AIF1 staining of non-immunological cells in normal adult tissues.
These findings are largely consistent with the RNA and protein data described in the Human Protein Atlas (Tissue expression AIF1)
Positive control: Colon: An at least moderate to strong cytoplasmic AIF1 immunostaining should be seen in mucosal macrophages.
Negative control: Colon: AIF1 immunostaining should be absent in all non-inflammatory cells.
Staining Pattern in Relevant Tumor Types
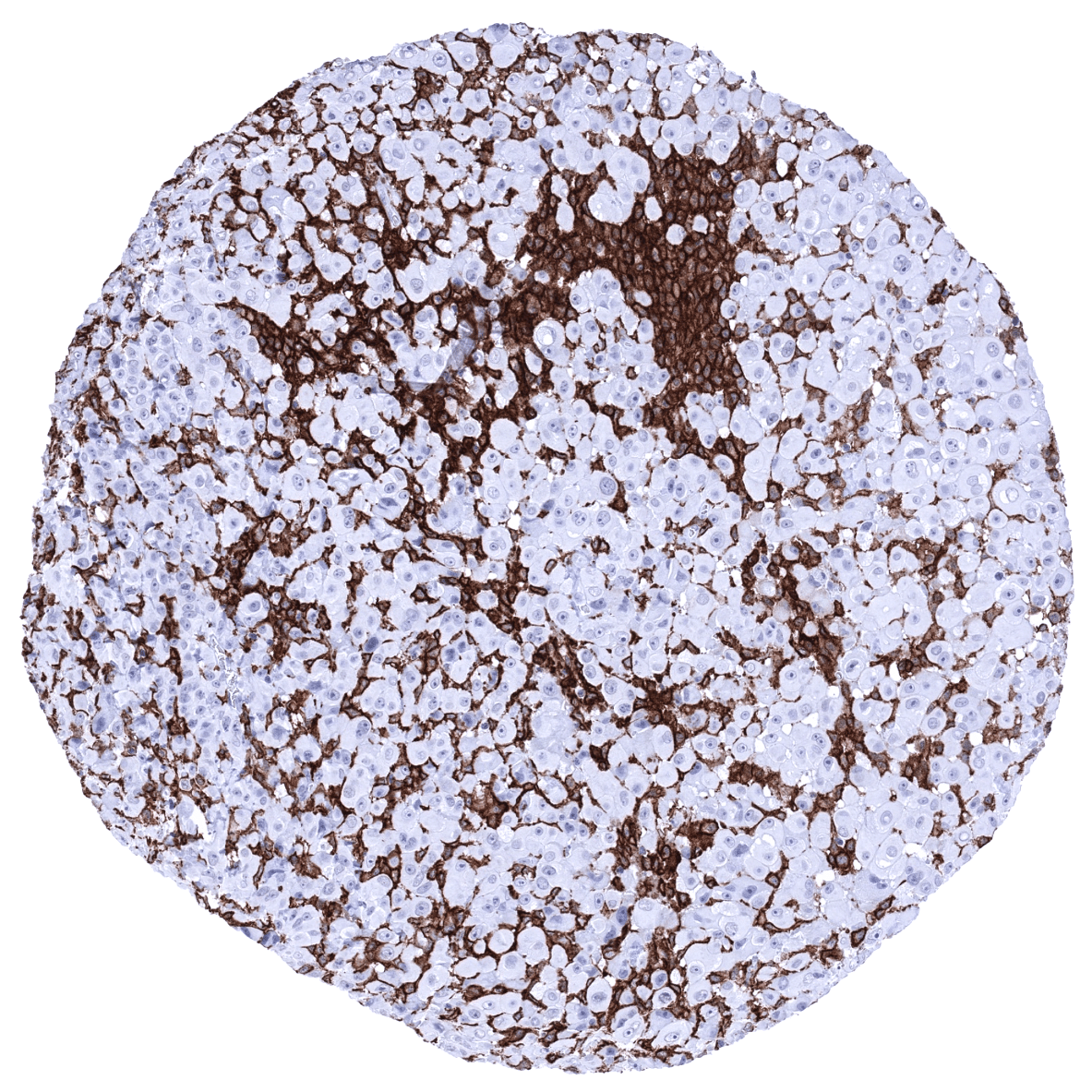
A positive AIF1 immunostaining of inflammatory cells is invariably seen in tumors. The quantity of positive cells and also the staining intensity of positive cells is highly variable, however. There is no evidence of AIF1 production in epithelial tumor cells so far. TCGA data suggest, that a high level of AIF1 RNA expression may be linked to unfavorable disease course in kidney cancer.
The TCGA findings on AIF1 RNA expression in different tumor categories have been summarized in the Human Protein Atlas.
Compatibility of Antibodies
No data available at the moment
Protocol Recommendations
IHC users have different preferences on how the stains should look like. Some prefer high staining intensity of the target stain and even accept some background. Others favor absolute specificity and lighter target stains. Factors that invariably lead to more intense staining include higher concentration of the antibody and visualization tools, longer incubation time, higher temperature during incubation, higher temperature and longer duration of the heat induced epitope retrieval (slide pretreatment). The impact of the pH during slide pretreatment has variable effects and depends on the antibody and the target protein.
All images and data shown here and in our image galleries are obtained by the manual protocol described below. Other protocols resulting in equivalent staining are described as well.
Manual protocol
Freshly cut sections should be used (less than 10 days between cutting and staining). Heat-induced antigen retrieval for 5 minutes in an autoclave at 121°C in pH 7,8 Target Retrieval Solution buffer. Apply MSVA-955M at a dilution of 1:150 at 37°C for 60 minutes. Visualization of bound antibody by the EnVision Kit (Dako, Agilent) according to the manufacturer’s directions.
Agilent / Dako – Autostainer Link 48
Pretreatment in PT-Link for 30 minutes at 95°C (pH high); FLEX peroxidase blocking for 5 minutes (room temperature), MSVA-955M 1:150 for 20 minutes (room temperature), FLEX+ mouse/rabbit (LINKER) for 15 minutes (room temperature), horseradish peroxidase (HRP) for 20 minutes (room temperature), FLEX DAB+Sub-Chromo for 10 minutes (room temperature), FLEX hematoxylin for 5 minutes (room temperature).
These images reflect stainings by the protocol described above. It is of note that a comparable staining result can also be obtained by different protocols. In general, a longer pretreatment, a longer incubation time of the primary antibody, a higher antibody concentration, and a longer incubation time of FLEX+LINKER result in stronger staining, potentially at the cost of more background staining. Modifications of the protocol with a strengthening effect on staining intensity in combination with changes of other parameters that result in lower staining intensity can result in a comparable result as shown above.
Leica – BOND RX
Dewax at 72°C for 30 seconds; Pretreatment in Bond Epitope Retrieval Solution (ER2 – EDTA pH9) for 20 minutes at 100°C; Peroxidase blocking for 5 minutes (room temperature), MSVA-955M 1:150 for 15 minutes (room temperature), Post primary (rabbit anti mouse) for 8 minutes (room temperature), Polymer (goat anti rabbit) for 8 minutes (room temperature), mixed DAB refine for 10 minutes (room temperature), hematoxylin for 5 minutes (room temperature).
These images reflect stainings by the protocol described above. It is of note that a comparable staining result can also be obtained by different protocols. In general, a longer pretreatment, a longer incubation time of the primary antibody, a higher antibody concentration, a higher temperature during incubation, and a longer incubation time of Post primary and or the Polymer result in stronger staining, potentially at the cost of more background staining. Modifications of the protocol with a strengthening effect on staining intensity in combination with changes of other parameters that result in lower staining intensity can result in a comparable result as shown above.
Roche – Ventana Discovery ULTRA
Pretreatment for 64 minutes at 100°C (pH 8,4); CM peroxidase blocking for 12 minutes (room temperature), MSVA-955M 1:150 for 20 minutes at 36°C, secondary antibody (anti-mouse HQ) for 12 minutes at 36°C, anti-HQ HRP for 12 minutes at room temperature, DAB at room temperature, hematoxylin II at room temperature for 8 minutes, bluing reagent at room temperature for 4 minutes.
These images depict staining results obtained by the protocol described above. It is of note, that the Ventana machines generally require higher antibody concentrations than other commonly used autostainers because the antibodies are automatically diluted during the procedure. Various other protocols can result in an identical result as shown above. A longer pretreatment, a longer incubation time of the primary antibody, a higher antibody concentration, a higher temperature during incubation, and a longer incubation time of secondary antibody and or the anti-HQ HRP result in stronger staining, potentially at the cost of more background staining.
Potential Research Applications
- AIF1 expression may represent a marker for the activity of macrophages. In general, AIF1 plays an important but largely unknown role in immunology. AIF1 is thus an important research target.
Evidence for Antibody Specificity in IHC
There are two ways how the specificity of an antibody can be documented for the application “immunohistochemistry on formalin fixed tissues”. These are: 1. comparison with a second independent method for target expression measurement across a large number of different tissue types (orthogonal strategy), and 2. Comparison with one or several independent antibodies for the same target and showing that all positive staining results are also seen with other antibodies for the same target (independent antibody strategy). Both methods were applied for MSVA-955M.
Orthogonal validation: For the antibody MSVA-955M, specificity is supported by the RNA expression data derived from three independent RNA screening studies, including the Human Protein Atlas (HPA) RNA-seq tissue dataset, the FANTOM5 project, and the Genotype-Tissue Expression (GTEx) project, which are all summarized in the Human Protein Atlas (Tissue expression AIF1). These databases describe a very high expression in lymphatic organs, macrophages, and granulocytes and a significant, but markedly lower RNA expression in all other organs. Such findings are highly consistent with expression of AIF1 in inflammatory cells only, as seen by using MSVA-955M.
Comparison of antibodies: In the human protein atlas, images are shown for the anti-AIF1 antibody HPA049234 which also show staining of histiocytes/macrophages and granulocytes in various different tissues. This further corroborates the specificity of AIF1 staining obtained by MSVA-955M. Most of all, HPA049234 also results in a membranous staining of glomeruli. This is a strong confirmation of the only AIF1 immunostaining obtained by MSVA-955M of non-immunological cells in normal adult tissues.