195,00 € – 695,00 €
Product details
Synonyms = Apomucin Major Airway Glycoprotein, Mucin 5 subtype AC tracheobronchial, Mucin 5 Subtypes A and C, Mucin 5AC oligomeric mucus/gel forming, Tracheobronchial Mucin
Antibody type = mouse monoclonal / IgG1
Clone = MSVA-109M
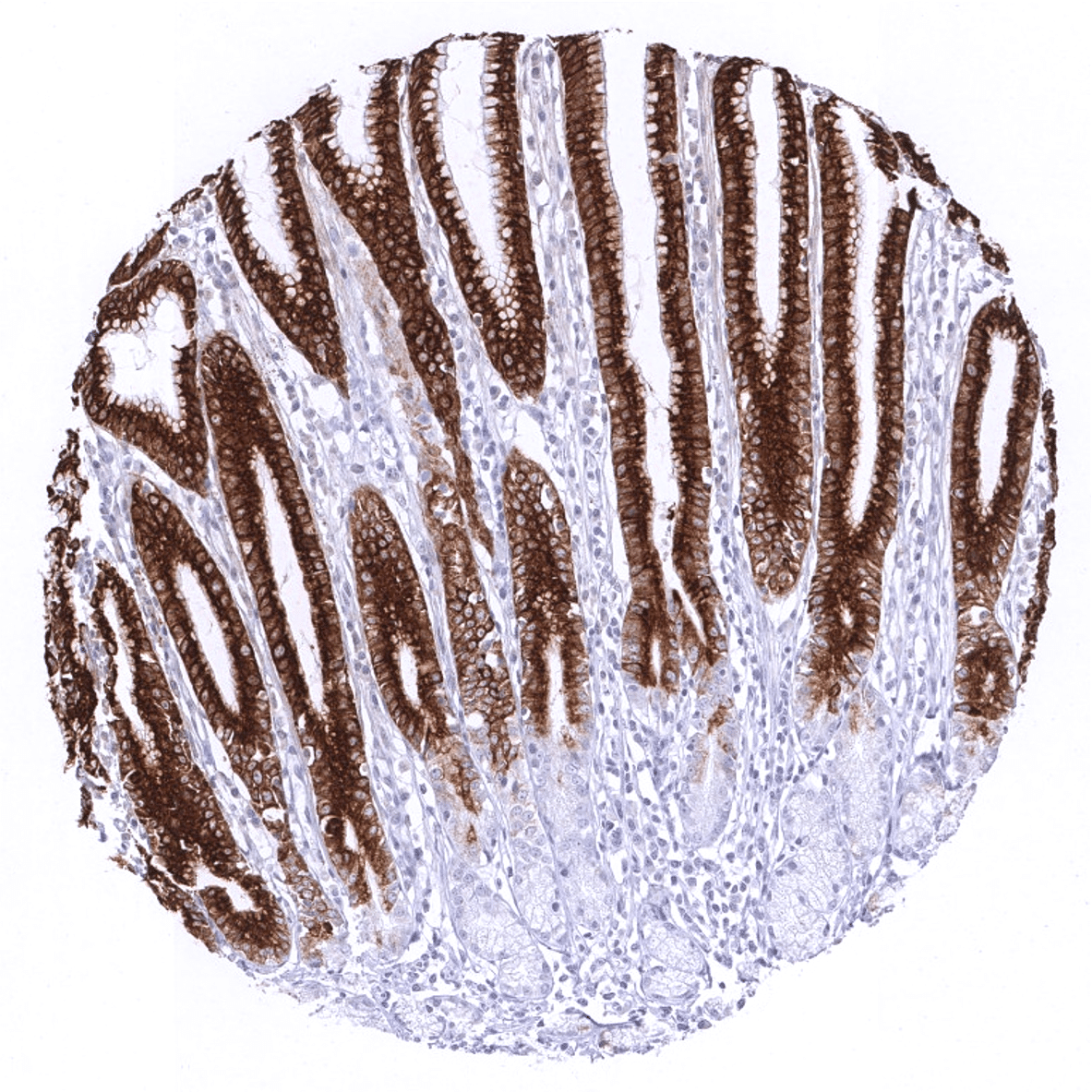
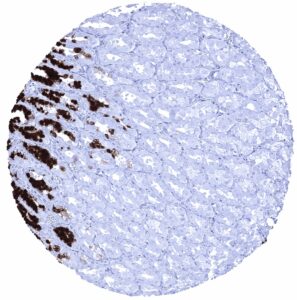
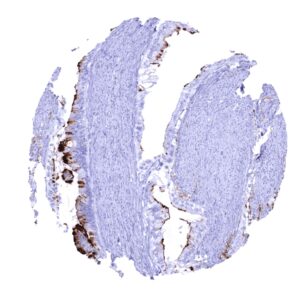
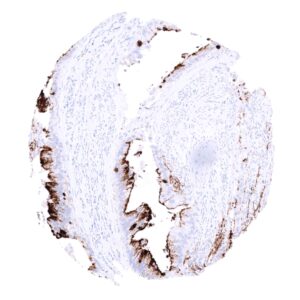
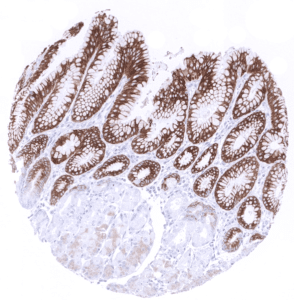
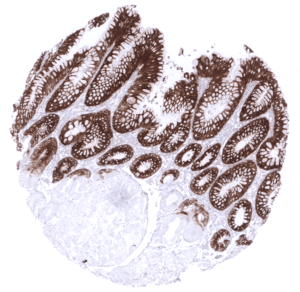
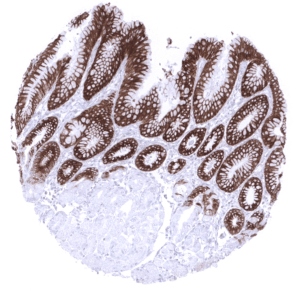
Positive control = Gastric mucosa: A strong cytoplasmic MUC5AC staining should be seen in the surface epithelium.
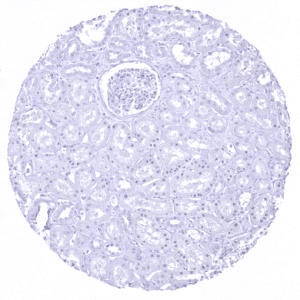
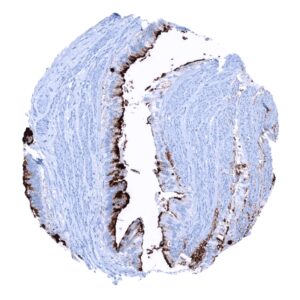
Negative control = Kidney: MUC5AC staining should be absent.
Cellular localization = Cytoplasmic
Reactivity = Human
Application = Immunohistochemistry
Dilution = 1:100 – 1:200
Intended Use = Research Use Only
Relevance of Antibody
Biology Behind
Mucin 5AC (MUC5AC) is one of five gel-forming mucins that are found in humans. It is encoded by the mucin cluster at chromosome 11p15, which also encodes the genes of the related mucins (MUC2, -5B, -6, and -19). The large (641 KDa) protein harbors multiple polymerization sites enabling formation of the mucin gel layer. Mucin layers are found at the surface of many epithelia, including those of the upper and lower respiratory tract, stomach, and endocervix, where they the build a barrier against chemical and mechanical damage. Microbial pathogens clotted in the mucin will be removed by the mucociliary system. Recent work suggests that MUC5AC may also play a role for tumor development and progression of lung, colorectal and pancreatic cancers, as it interacts with tumor relevant proteins such as p53, integrin ß4 and ß-catenin. In colorectal cancer, MUC5AC expression is independently linked to both right colon location and microsatellite instability.
Staining Pattern in Normal Tissues
MUC5AC staining pattern in Normal Tissues with antibody MSVA-109M (images are shown in our “Normal Tissue Gallery”)
| Brain | Cerebrum | Negative. |
| Cerebellum | Negative. | |
| Endocrine Tissues | Thyroid | Negative. |
| Parathyroid | Negative. | |
| Adrenal gland | Negative. | |
| Pituitary gland | Negative. | |
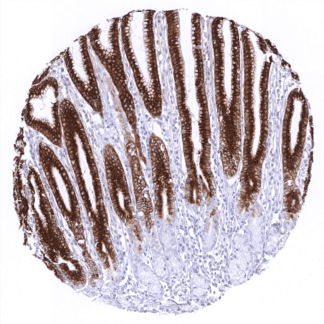
| Respiratory system | Respiratory epithelium | Strong cytoplasmic staining of goblet cells. |
| Lung | Negative. | |
| Gastrointestinal Tract | Salivary glands | Negative. |
| Esophagus | Negative. | |
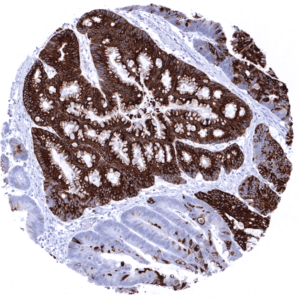
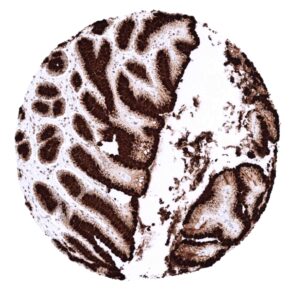
| Stomach | Strong cytoplasmic MUC5AC staining in all cells of the stomach surface epithelium. | |
| Colon | Few scattered MUC5AC positive cells do occur. | |
| Duodenum | Few scattered MUC5AC positive cells do occur. | |
| Rectum | Few scattered MUC5AC positive cells do occur. | |
| Small intestine | Few scattered MUC5AC positive cells do occur. | |
| Liver | Negative. | |
| Gallbladder | Moderate to strong cytoplasmic MUC5AC staining of the surface epithelium may be focal. | |
| Pancreas | Negative. | |
| Genitourinary | Kidney | Negative. |
| Urothelium | Negative. | |
| Male genital | Prostate | Negative. |
| Seminal vesicles | Negative. | |
| Testis | Negative. | |
| Epididymis | Negative. | |
| Female genital | Breast | Negative. |
| Uterus, ectocervix | Moderate to strong MUC5AC staining of some endocervical glands from some samples. | |
| Uterus endocervix | Negative. | |
| Uterus, endometrium | Negative. | |
| Fallopian Tube | Negative. | |
| Ovary | Negative. | |
| Placenta early | Negative. | |
| Placenta mature | Negative. | |
| Amnion | Negative. | |
| Chorion | Negative. | |
| Skin | Epidermis | Negative. |
| Sebaceous glands | Negative. | |
| Muscle/connective tissue | Heart muscle | Negative. |
| Skeletal muscle | Negative. | |
| Smooth muscle | Negative. | |
| Fat | Negative. | |
| Bone marrow/lymphoid | Bone marrow | Negative. |
| Lymph node | Negative. | |
| Spleen | Negative. | |
| Thymus | Negative. | |
| Tonsil | Negative. | |
| Remarks | MUC5AC staining is always cytoplasmic. It is predominantly expressed in the surface epithelium of the stomach and in goblet cells of the respiratory tract. |
These findings are largely consistent with the RNA and protein data described in the Human Protein Atlas (Tissue expression MUC5AC)
Suggested positive tissue control: Gastric mucosa: A strong cytoplasmic MUC5AC staining should be seen in the surface epithelium.
Suggested negative tissue control: Kidney: MUC5AC staining should be absent.
Staining Pattern in Relevant Tumor Types
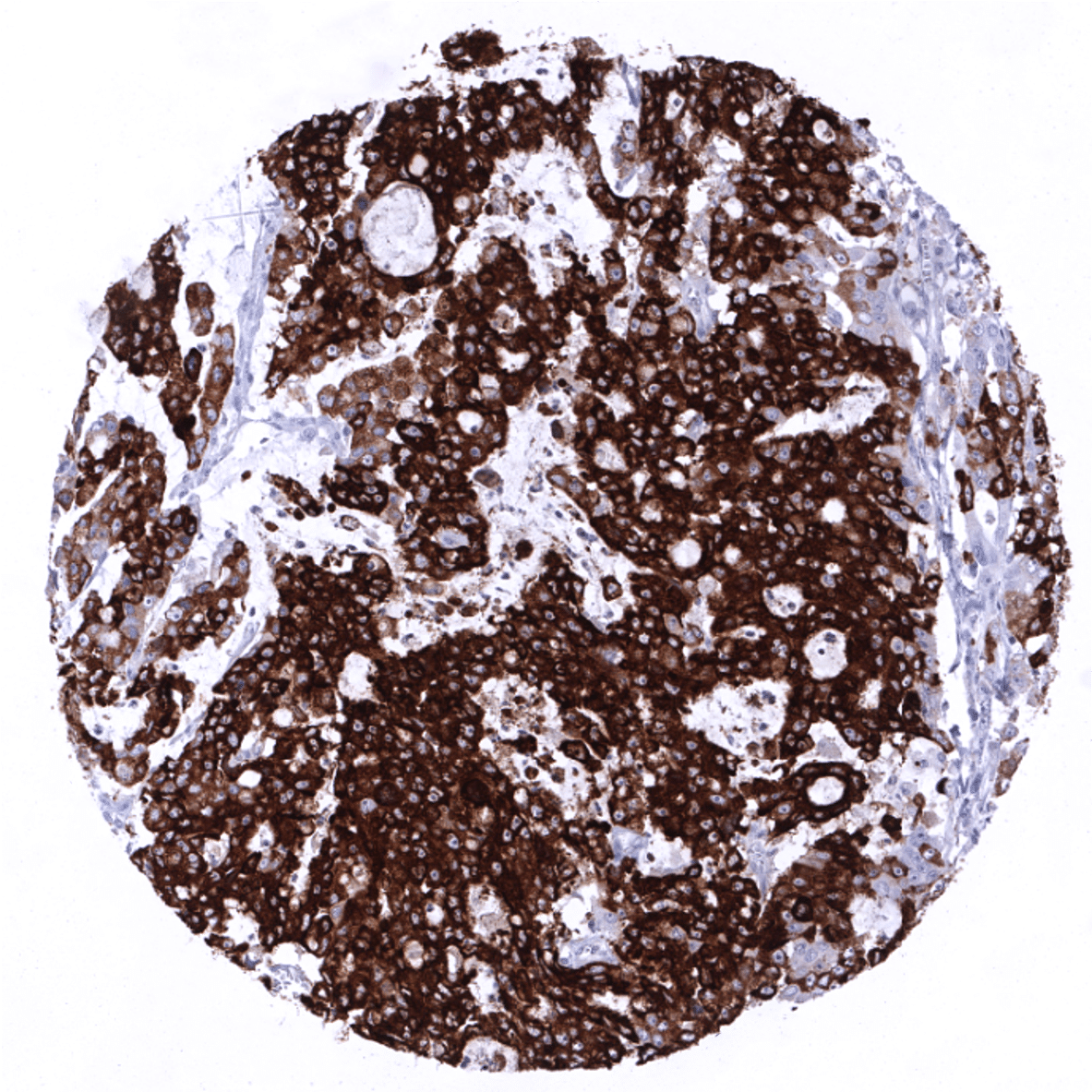
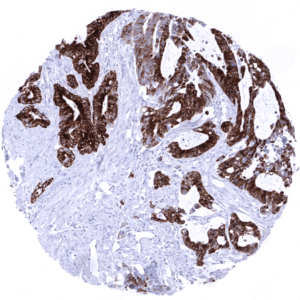
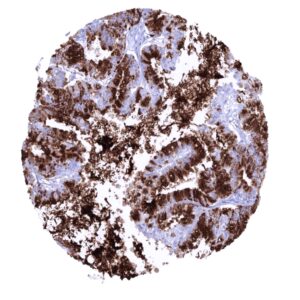
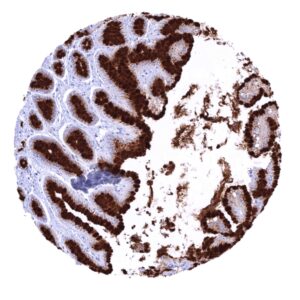
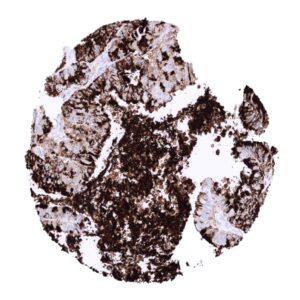
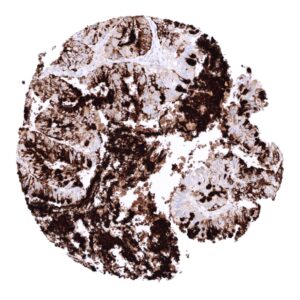
In principle, three different patterns can be seen: diffuse staining of all cells, patchy focal staining of tumor areas that sometimes exhibit a different morphology as compared to non-stained areas groups, and a mosaic pattern containing a variable number of scattered strongly MUC5AC positive cancer cells quite regularly distributed between clearly negative tumor cells. MUC5AC expression is most commonly seen in adenocarcinomas derived from esophagus (>70%), pancreas (>60%), and the stomach (>40%). At a frequency below 30% it is also seen in various other adenocarcinomas such as endometrial cancer, colorectal carcinoma,cholangiocellular carcinoma, and lung adenocarcinoma. At a frequency below 15% MUC5AC positivity is also seen in various other tumor types including squamous cell carcinoma of the lung, neuroendocrine cancer, large cell lung cancer. MUC5AC is rarely or never present in various important other tumor types such as in cancers of the breast, prostate, kidney, serous ovarian cancer, hepatocellular carcinoma, or malignant mesothelioma.
The TCGA findings on MUC5AC RNA expression in different tumor categories have been summarized in the Human Protein Atlas.
Compatibility of Antibodies
MUC5AC (MSVA-109M) publication summary
Papers used for data compilation:
- Rico SD et al. “MUC5AC Expression in Various Tumor Types and Nonneoplastic Tissue: A Tissue Microarray Study on 10 399 Tissue Samples.” Published in Technol Cancer Res Treat. Jan-Dec 2021 PMID: 34547930
- Rico SD et al. “MUC5AC expression is linked to mucinous/endometroid subtype, absence of nodal metastasis and mismatch repair deficiency in ovarian cancer.” Published in Pathol Res Pract. 2021 Aug; 224:153533 PMID: 34171599
- Rico SD et al. “Elevated MUC5AC expression is associated with mismatch repair deficiency and proximal tumor location but not with cancer progression in colon cancer.” Published in Med Mol Morphol. 2021 Jun;54(2): 156-165 PMID: 33373033
In these three studies, a total of 10210 tumors were analyzed from 114 different tumor categories by using the following protocol: Heat-induced antigen retrieval for 5 minutes in an autoclave at 121°C in pH 7.8 Target Retrieveal Solution buffer. MSVA-109M at a dilution of 1:150 at 37°C for 60 minutes. Visualization of bound antibody by the EnVision Kit (Dako, Agilent).This protocol was also used for all stainings depicted in our tumor and normal tissue galleries.
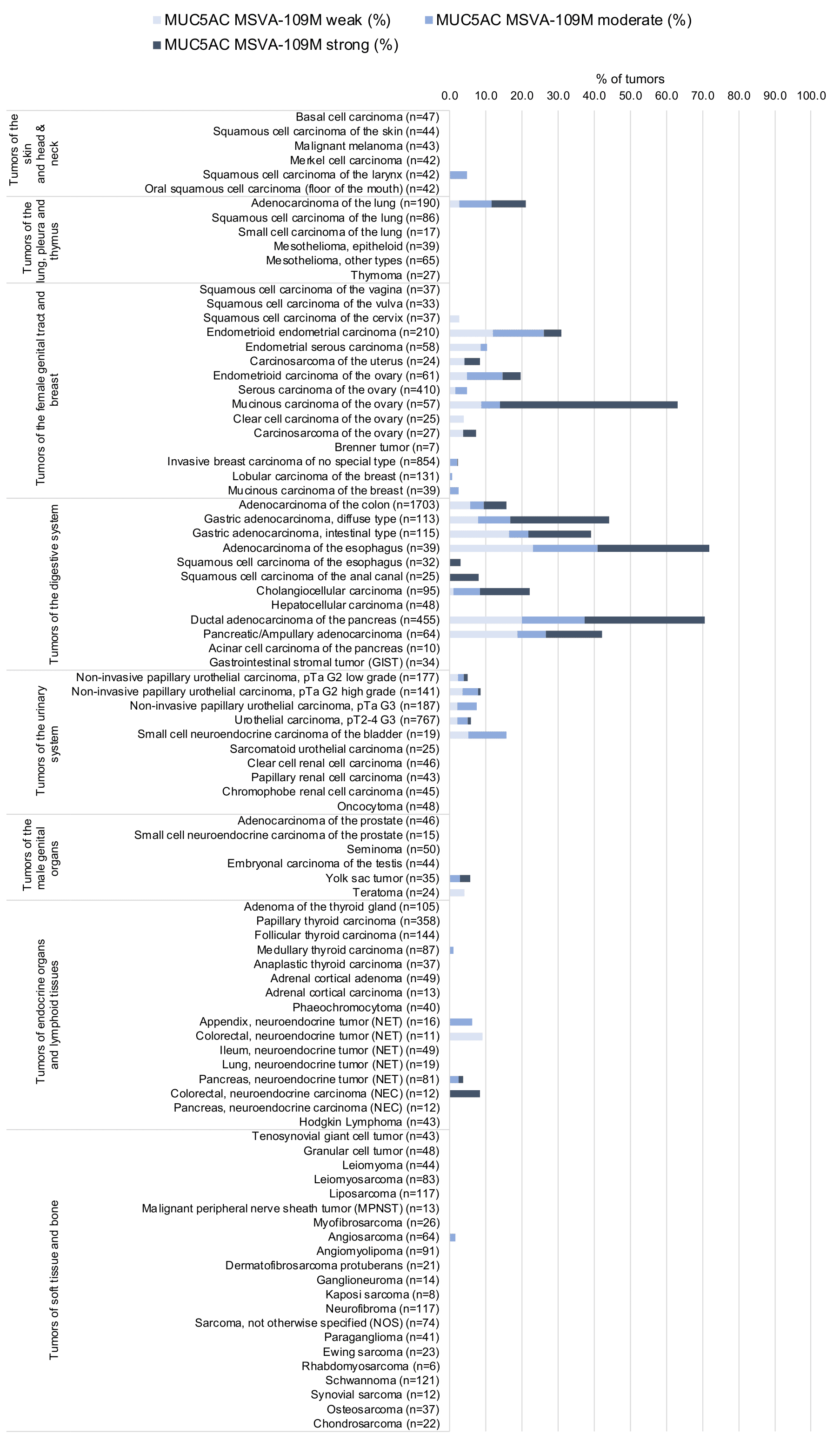
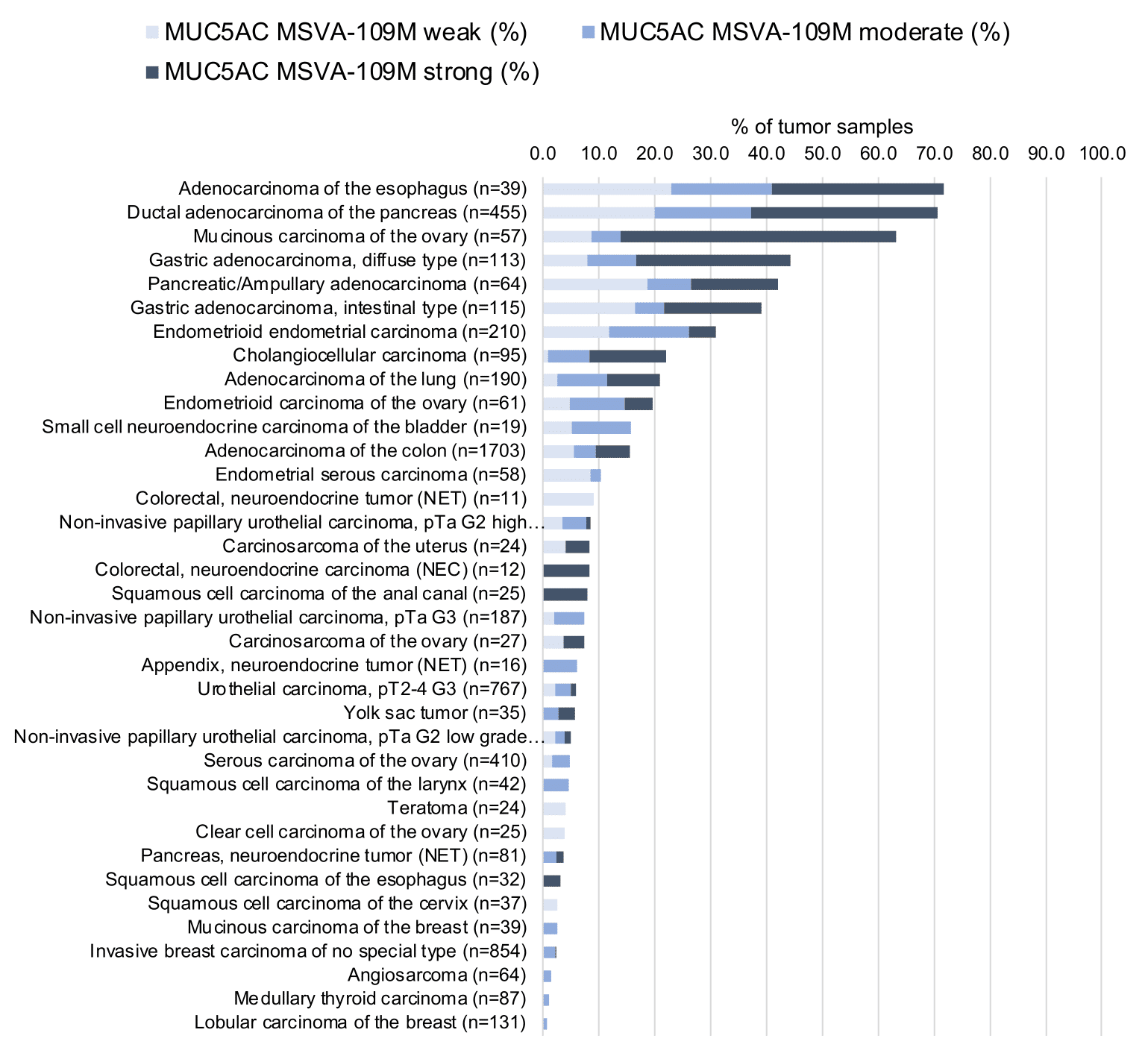
At least one case with a positive MUC5AC immunostaining was seen in 47 (41,2%) and at least one case with a strong MUC5AC immunostaining was seen in 30 (26,3%) of 114 tumor categories. The distribution of positive staining results is shown in an “organ-systematic” and in a “ranking order” figure below (images based on a compilation of data from Rico et al.). Results on possible associations with histopathological and clinical parameters of tumor aggressiveness are also summarized below (table based on data from Rico et al.).
Authors conclusions on diagnostic utility with respect to the distinction of benign versus malignant (Rico SD et al.):
- In organs where MUC5AC expression is lacking in normal cells, a positive MUC5AC immunostaining may serve as a useful surrogate marker of neoplastic transformation. For example, the nonneoplastic pancreas does not express MUC5AC, while MUC5AC expression can be detected in most pancreatic adenocarcinomas and even in its precursor lesions as early as low-grade PanINs.
Authors conclusions on diagnostic utility with respect to the distinction of different tumor entities (Rico SD et al.):
- A positive MUC5AC immunostaining might be considered an argument against a tumor origin from organs that never or very rarely gave rise to MUC5AC positive cancer cells, such as the breast, prostate, and the kidney.
Authors conclusions on prognostic/predictive role of PLAP expression (Rico SD et al.):
- The level of MUC5AC expression is unrelated to parameters of cancer aggressiveness in colorectal cancer.
- A marginal statistical association was observed between MUC5AC expression and absence of nodal metastasis in endometroid and serous ovarian cancer.
Data from the publication by Rico SD et al. “MUC5AC Expression in Various Tumor Types and Nonneoplastic Tissue: A Tissue Microarray Study on 10 399 Tissue Samples.” Published in Technol Cancer Res Treat. Jan-Dec 2021 PMID: 34547930 Summarized in own graphics:
1. MUC5AC staining in tumors “organ-specific” with antibody MSVA-109M
2. MUC5AC staining in tumors “ranking order” by positivity with antibody MSVA-109M.
Protocol Recommendations
IHC users have different preferences on how the stains should look like. Some prefer high staining intensity of the target stain and even accept some background. Others favor absolute specificity and lighter target stains. Factors that invariably lead to more intense staining include higher concentration of the antibody and visualization tools, longer incubation time, higher temperature during incubation, higher temperature and longer duration of the heat induced epitope retrieval (slide pretreatment). The impact of the pH during slide pretreatment has variable effects and depends on the antibody and the target protein. Accordingly, multiple different protocols can generate identical staining results.
All images and data shown here are and in our image galleries were obtained by the manual protocol described below. Other protocols resulting in equivalent staining are described as well.
Manual protocol
Freshly cut sections should be used (less than 10 days between cutting and staining). Heat-induced antigen retrieval for 5 minutes in an autoclave at 121°C in pH 7.8 Target Retrieveal Solution buffer. Apply MSVA-109M at a dilution of 1:150 at 37°C for 60 minutes. Visualization of bound antibody by the EnVision Kit (Dako, Agilent) according to the manufacturer’s directions.
Agilent / Dako – Autostainer Link 48
Pretreatment in PT-Link for 30 minutes at 95°C (pH high); FLEX peroxidase blocking for 5 minutes (room temperature), MSVA-109M 1:150 for 20 minutes (room temperature), FLEX+ mouse/rabbit (LINKER) for 15 minutes (room temperature), horseradish peroxidase (HRP) for 20 minutes (room temperature), FLEX DAB+Sub-Chromo for 10 minutes (room temperature), FLEX hematoxylin for 5 minutes (room temperature).
These images reflect stainings by the protocol described above. It is of note that a comparable staining result can also be obtained by different protocols. In general, a longer pretreatment, a longer incubation time of the primary antibody, a higher antibody concentration, and a longer incubation time of FLEX+LINKER result in stronger staining, potentially at the cost of more background staining. Modifications of the protocol with a strengthening effect on staining intensity in combination with changes of other parameters that result in lower staining intensity can result in a comparable result as shown above.
Leica – BOND RX
Dewax at 72°C for 30 seconds; Pretreatment in Bond Epitope Retrieval Solution (ER2 – EDTA pH9) for 20 minutes at 100°C; Peroxidase blocking for 5 minutes (room temperature), MSVA-109M 1:150 for 15 minutes (room temperature), Post primary (rabbit anti mouse) for 8 minutes (room temperature), Polymer (goat anti rabbit) for 8 minutes (room temperature), mixed DAB refine for 10 minutes (room temperature), hematoxylin for 5 minutes (room temperature).
These images reflect stainings by the protocol described above. It is of note that a comparable staining result can also be obtained by different protocols. In general, a longer pretreatment, a longer incubation time of the primary antibody, a higher antibody concentration, a higher temperature during incubation, and a longer incubation time of Post primary and or the Polymer result in stronger staining, potentially at the cost of more background staining. Modifications of the protocol with a strengthening effect on staining intensity in combination with changes of other parameters that result in lower staining intensity can result in a comparable result as shown above.
Roche – Ventana Discovery ULTRA
Pretreatment for 64 minutes at 100°C (pH 8,4); CM peroxidase blocking for 12 minutes (room temperature), MSVA-109M 1:150 for 20 minutes at 36°C, secondary antibody (anti-mouse HQ) for 12 minutes at 36°C, anti-HQ HRP for 12 minutes at room temperature, DAB at room temperature, hematoxylin II at room temperature for 8 minutes, bluing reagent at room temperature for 4 minutes.
These images depict staining results obtained by the protocol described above. It is of note, that the Ventana machines generally require higher antibody concentrations than other commonly used autostainers because the antibodies are automatically diluted during the procedure. Various other protocols can result in an identical result as shown above. A longer pretreatment, a longer incubation time of the primary antibody, a higher antibody concentration, a higher temperature during incubation, and a longer incubation time of secondary antibody and or the anti-HQ HRP result in stronger staining, potentially at the cost of more background staining.
Impact of pH
The strongest MUC5AC staining by MSVA-109M is obtained at a pH 9,0. However, pH 7,8 results in only a slight reduction of the staining intensity as compared to pH9. We thus consider pH7,8 as optimal for manual staining because of the better tissue preservation at pH7,8 than at pH 9,0.
Potential Research Applications
- A comprehensive study analyzing MUC5AC expression in various different tumor entities would be helpful to assess the diagnostic significance of MUC5AC IHC.
- The prognostic relevance of MUC5AC expression and of different immunostaining patterns (diffuse vs “mosaic”) should be evaluated.
- The use of serum MUC5AC measurement as a putative instrument for monitoring response to therapy and early detection of recurrence in case of MUC5AC positive primary tumors should be investigated.
Evidence for Antibody Specificity in IHC
Specificity of MSVA-109M is documented by strong positive staining in cell types that are well documented to express MUC5AC such as surface epithelium of the stomach and bronchial epithelium goblet cells and absence of staining in all tissues known to not express MUC5AC including tissues notorious for non-specific IHC background such as kidney, colonic mucosa, and epidermis.